QUESTION IMAGE
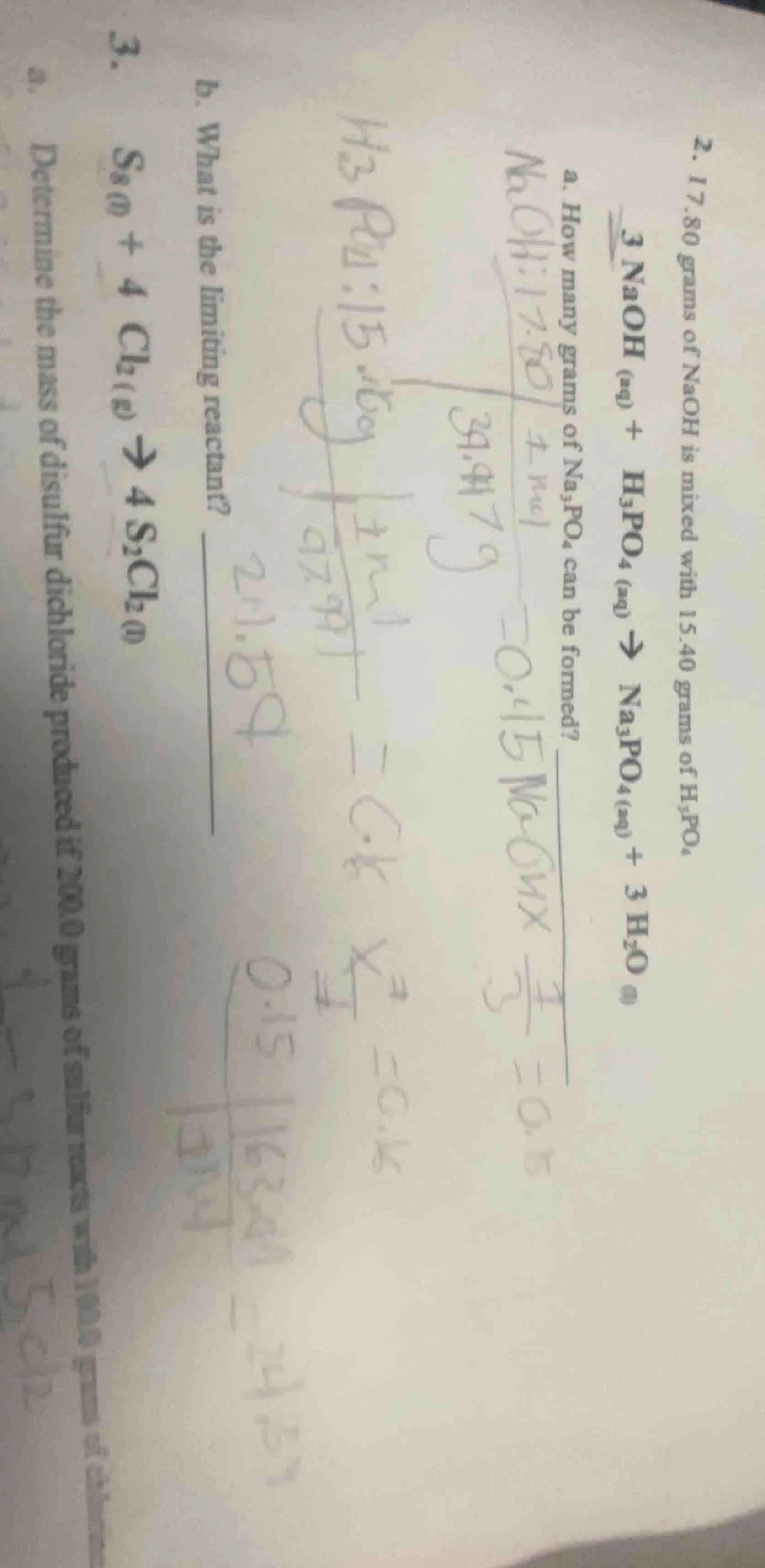
Question
- 17.80 grams of naoh is mixed with 15.40 grams of $h_3po_4$$3naoh_{(aq)} + h_3po_{4(aq)} \
ightarrow na_3po_{4(aq)} + 3h_2o_{(l)}$a. how many grams of $na_3po_4$ can be formed?b. what is the limiting reactant?3. $s_{8(l)} + 4cl_{2(g)} \
ightarrow 4s_2cl_{2(l)}$a. determine the mass of disulfur dichloride produced if 200.0 grams of sulfur reacts with 100.0 grams of chlorine
Step1: Calculate moles of NaOH
Molar mass of NaOH = $23 + 16 + 1 = 40$ g/mol
$\text{Moles of NaOH} = \frac{17.80\ \text{g}}{40\ \text{g/mol}} = 0.445\ \text{mol}$
Step2: Calculate moles of $\text{H}_3\text{PO}_4$
Molar mass of $\text{H}_3\text{PO}_4 = 3\times1 + 31 + 4\times16 = 98$ g/mol
$\text{Moles of }\text{H}_3\text{PO}_4 = \frac{15.40\ \text{g}}{98\ \text{g/mol}} \approx 0.1571\ \text{mol}$
Step3: Find limiting reactant
From reaction: $3\text{NaOH} + \text{H}_3\text{PO}_4
ightarrow \text{Na}_3\text{PO}_4 + 3\text{H}_2\text{O}$
Mole ratio of $\text{NaOH}:\text{H}_3\text{PO}_4 = 3:1$
Moles of $\text{H}_3\text{PO}_4$ needed for NaOH: $\frac{0.445}{3} \approx 0.1483\ \text{mol}$
We have 0.1571 mol of $\text{H}_3\text{PO}_4$, which is more than needed. So NaOH is limiting.
Step4: Calculate moles of $\text{Na}_3\text{PO}_4$
Mole ratio of $\text{NaOH}:\text{Na}_3\text{PO}_4 = 3:1$
$\text{Moles of }\text{Na}_3\text{PO}_4 = \frac{0.445}{3} \approx 0.1483\ \text{mol}$
Step5: Calculate mass of $\text{Na}_3\text{PO}_4$
Molar mass of $\text{Na}_3\text{PO}_4 = 3\times23 + 31 + 4\times16 = 164$ g/mol
$\text{Mass of }\text{Na}_3\text{PO}_4 = 0.1483\ \text{mol} \times 164\ \text{g/mol} \approx 24.32\ \text{g}$
---
Step6: Calculate moles of $\text{S}_8$ (Q3a)
Molar mass of $\text{S}_8 = 8\times32 = 256$ g/mol
$\text{Moles of }\text{S}_8 = \frac{200.0\ \text{g}}{256\ \text{g/mol}} \approx 0.78125\ \text{mol}$
Step7: Calculate moles of $\text{Cl}_2$ (Q3a)
Molar mass of $\text{Cl}_2 = 2\times35.45 = 70.9$ g/mol
$\text{Moles of }\text{Cl}_2 = \frac{100.0\ \text{g}}{70.9\ \text{g/mol}} \approx 1.410\ \text{mol}$
Step8: Find limiting reactant (Q3a)
Reaction: $\text{S}_8 + 4\text{Cl}_2
ightarrow 4\text{S}_2\text{Cl}_2$
Mole ratio $\text{S}_8:\text{Cl}_2 = 1:4$
Moles of $\text{Cl}_2$ needed for $\text{S}_8$: $0.78125 \times 4 = 3.125\ \text{mol}$
We only have 1.410 mol $\text{Cl}_2$, so $\text{Cl}_2$ is limiting.
Step9: Calculate mass of $\text{S}_2\text{Cl}_2$ (Q3a)
Mole ratio $\text{Cl}_2:\text{S}_2\text{Cl}_2 = 4:4 = 1:1$
$\text{Moles of }\text{S}_2\text{Cl}_2 = 1.410\ \text{mol}$
Molar mass of $\text{S}_2\text{Cl}_2 = 2\times32 + 2\times35.45 = 135.9$ g/mol
$\text{Mass of }\text{S}_2\text{Cl}_2 = 1.410\ \text{mol} \times 135.9\ \text{g/mol} \approx 191.6\ \text{g}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2a. $\approx 24.3$ grams of $\text{Na}_3\text{PO}_4$ can be formed
2b. The limiting reactant is $\text{NaOH}$
3a. $\approx 192$ grams of disulfur dichloride is produced
3b. The limiting reactant is $\text{Cl}_2$