QUESTION IMAGE
Question
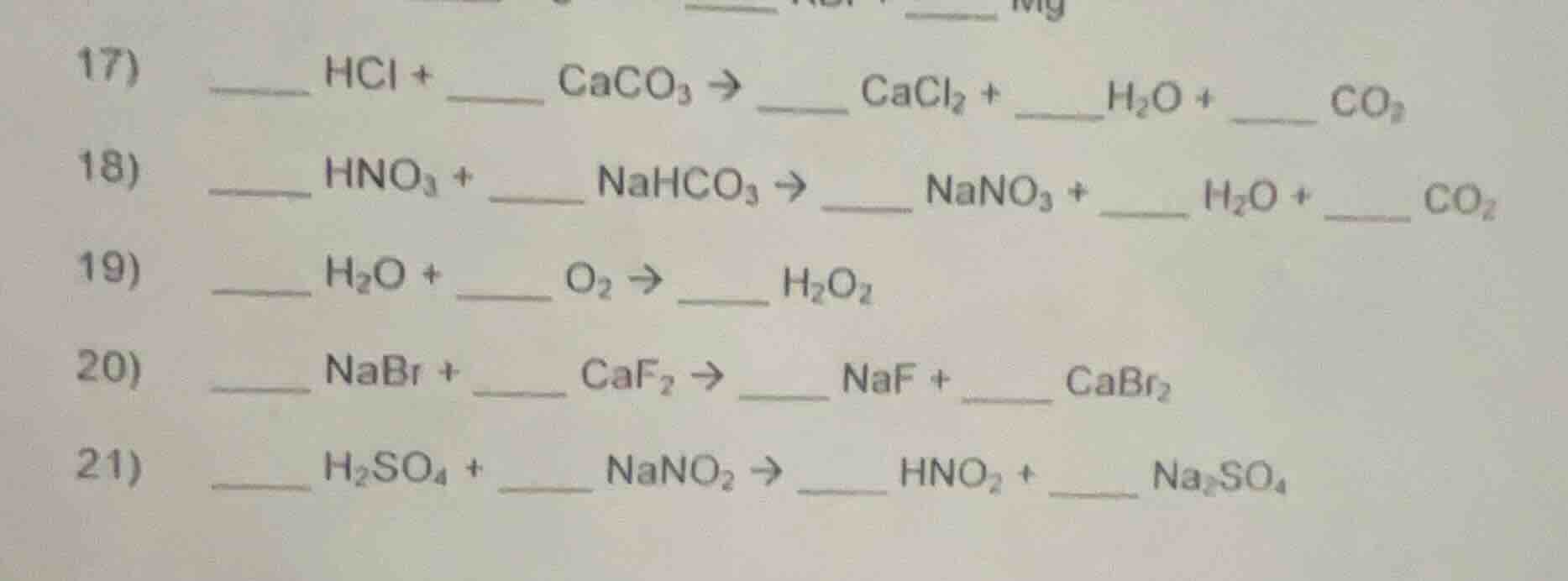
17)
____ hcl + __ caco₃ → __ cacl₂ + __h₂o + ____ co₂
18)
____ hno₃ + __ nahco₃ → __ nano₃ + __ h₂o + ____ co₂
19)
____ h₂o + __ o₂ → ____ h₂o₂
20)
____ nabr + __ caf₂ → __ naf + ____ cabr₂
21)
____ h₂so₄ + __ nano₂ → __ hno₂ + ____ na₂so₄
Problem 17:
Step1: Balance Cl.
On the right, \( \text{CaCl}_2 \) has 2 Cl. So \( \text{HCl} \) needs coefficient 2.
\( 2\text{HCl} + \text{CaCO}_3
ightarrow \text{CaCl}_2 + \text{H}_2\text{O} + \text{CO}_2 \)
Step2: Check other atoms.
Ca: 1 on both sides. C: 1 on both sides. O: 3 in \( \text{CaCO}_3 \), 1 in \( \text{H}_2\text{O} \) and 2 in \( \text{CO}_2 \) (total 3). H: 2 in \( \text{HCl} \) and 2 in \( \text{H}_2\text{O} \). Balanced.
Step1: Balance Na.
\( \text{NaHCO}_3 \) and \( \text{NaNO}_3 \) have 1 Na each. So coefficients for \( \text{NaHCO}_3 \) and \( \text{NaNO}_3 \) are 1.
\( \text{HNO}_3 + 1\text{NaHCO}_3
ightarrow 1\text{NaNO}_3 + \text{H}_2\text{O} + \text{CO}_2 \)
Step2: Balance H, N, C, O.
H: 1 in \( \text{HNO}_3 \) and 1 in \( \text{NaHCO}_3 \) (total 2), 2 in \( \text{H}_2\text{O} \). N: 1 on both sides. C: 1 on both sides. O: 3 in \( \text{HNO}_3 \), 3 in \( \text{NaHCO}_3 \) (total 6), 3 in \( \text{NaNO}_3 \), 1 in \( \text{H}_2\text{O} \), 2 in \( \text{CO}_2 \) (total 6). Balanced.
Step1: Balance O.
Left: \( \text{H}_2\text{O} \) has 1 O, \( \text{O}_2 \) has 2 O (total 1 + 2x). Right: \( \text{H}_2\text{O}_2 \) has 2 O (total 2y). Let x=0.5, y=1. Multiply by 2 to eliminate fractions.
\( 2\text{H}_2\text{O} + 1\text{O}_2
ightarrow 2\text{H}_2\text{O}_2 \)
Step2: Check H.
Left: 4 H, Right: 4 H. O: 21 + 2 = 4, Right: 22 = 4. Balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2, 1, 1, 1, 1