QUESTION IMAGE
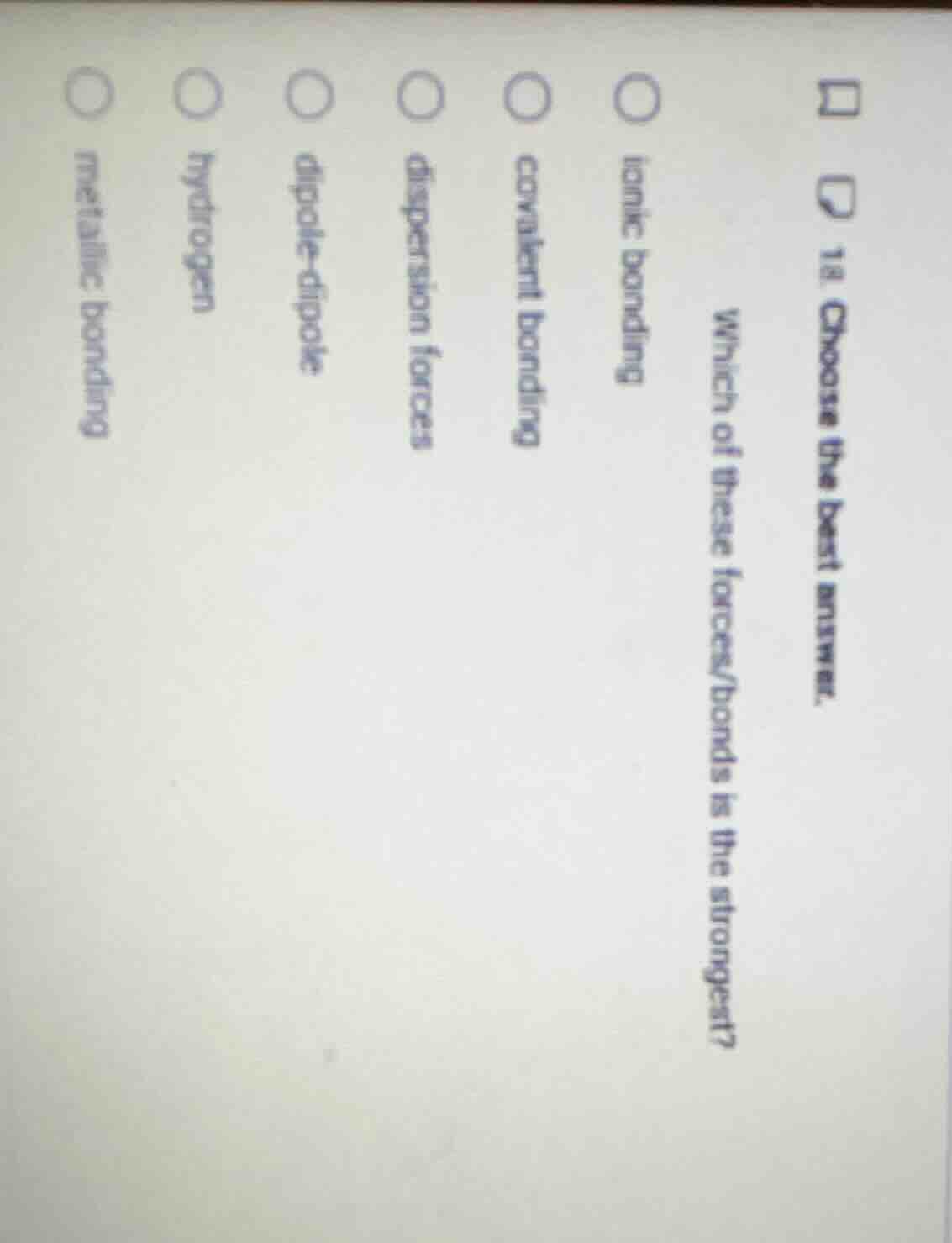
Question
- choose the best answer. which of these forces/bonds is the strongest?
ionic bonding
covalent bonding
dispersion forces
dipole-dipole
hydrogen
metallic bonding
Brief Explanations
Intermolecular forces (dispersion, dipole-dipole, hydrogen) are weaker than intramolecular bonds. Among intramolecular bonds, ionic bonding involves electrostatic attraction between oppositely charged ions, which is stronger than covalent (shared electrons) and metallic (delocalized electrons) bonding due to the full charge separation.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. ionic bonding