QUESTION IMAGE
Question
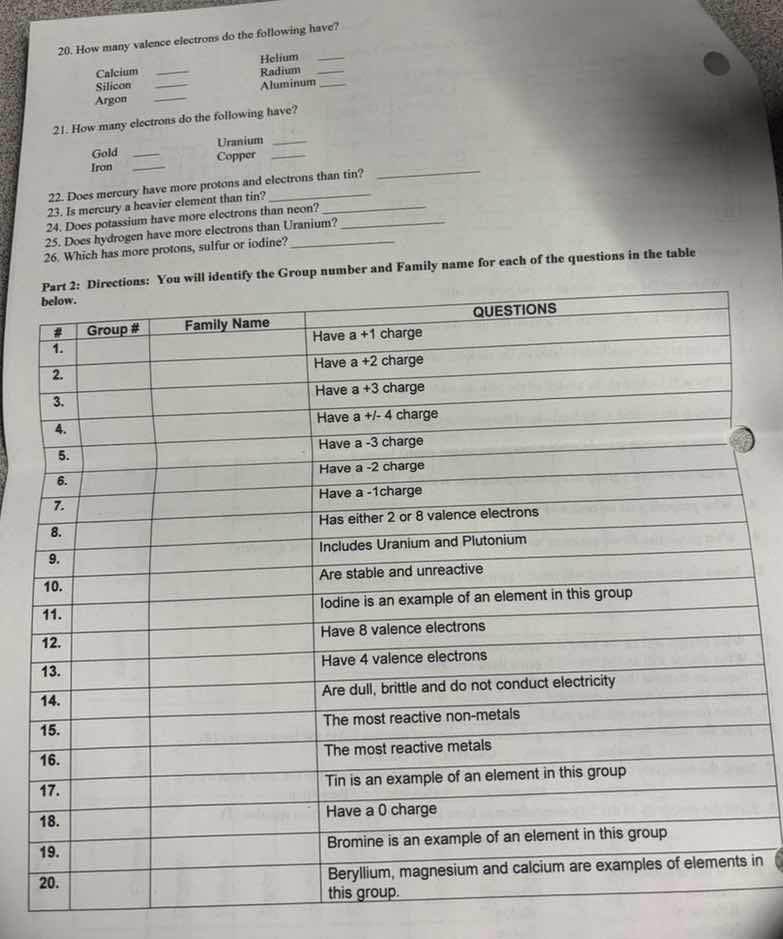
- how many valence electrons do the following have?calcium ____ helium __silicon __ radium __argon __ aluminum __21. how many electrons do the following have?gold __ uranium __iron __ copper __22. does mercury have more protons and electrons than tin? ___________23. is mercury a heavier element than tin? ___________24. does potassium have more electrons than neon? ___________25. does hydrogen have more electrons than uranium? ___________26. which has more protons, sulfur or iodine? _____________part 2: directions: you will identify the group number and family name for each of the questions in the table below.# | group # | family name | questions1. | | | have a +1 charge2. | | | have a +2 charge3. | | | have a +3 charge4. | | | have a +/-4 charge5. | | | have a -3 charge6. | | | have a -2 charge7. | | | have a -1 charge8. | | | has either 2 or 8 valence electrons9. | | | includes uranium and plutonium10. | | | are stable and unreactive11. | | | iodine is an example of an element in this group12. | | | have 8 valence electrons13. | | | have 4 valence electrons14. | | | are dull, brittle and do not conduct electricity15. | | | the most reactive non-metals16. | | | the most reactive metals17. | | | tin is an example of an element in this group18. | | | have a 0 charge19. | | | bromine is an example of an element in this group20. | | | beryllium, magnesium and calcium are examples of elements in this group
Step1: Find valence electrons (Group A)
Valence electrons = Group A number (main groups); He = 2.
- Calcium (Group 2A): 2
- Helium (Group 18): 2
- Silicon (Group 4A): 4
- Radium (Group 2A): 2
- Argon (Group 18): 8
- Aluminum (Group 3A): 3
Step2: Find total electrons (neutral = atomic #)
Total electrons = Atomic number (neutral atoms):
- Gold (Atomic #79): 79
- Uranium (Atomic #92): 92
- Iron (Atomic #26): 26
- Copper (Atomic #29): 29
Step3: Compare protons/electrons (atomic # = protons = electrons)
- Q22: Hg (#80) > Sn (#50): Yes
- Q23: Hg (200.59 amu) > Sn (118.71 amu): Yes
- Q24: K (#19) < Ne (#10)? No (19>10): Yes
- Q25: H (#1) < U (#92): No
- Q26: S (#16) < I (#53): Iodine
Step4: Match groups/families to properties
- Group 1, Alkali Metals (+1 charge)
- Group 2, Alkaline Earth Metals (+2 charge)
- Group 13, Boron Group (+3 charge)
- Group 14, Carbon Group (+/-4 charge)
- Group 15, Nitrogen Group (-3 charge)
- Group 16, Chalcogens (-2 charge)
- Group 17, Halogens (-1 charge)
- Groups 2,18 (2/8 valence)
- Group 3-12, Transition Metals (includes U, Pu)
- Group 18, Noble Gases (stable/unreactive)
- Group 17, Halogens (Iodine is member)
- Group 18, Noble Gases (8 valence)
- Group 14, Carbon Group (4 valence)
- Nonmetals (Groups 14-18, except noble gases)
- Group 17, Halogens (most reactive nonmetals)
- Group 1, Alkali Metals (most reactive metals)
- Group 14, Carbon Group (Tin is member)
- Group 18, Noble Gases (0 charge)
- Group 17, Halogens (Bromine is member)
- Group 2, Alkaline Earth Metals (Be, Mg, Ca members)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Part 1
- Valence electrons:
Calcium: 2; Helium: 2; Silicon: 4; Radium: 2; Argon: 8; Aluminum: 3
- Total electrons:
Gold: 79; Uranium: 92; Iron: 26; Copper: 29
- Yes
- Yes
- Yes
- No
- Iodine
Part 2
| # | Group # | Family Name |
|---|---|---|
| 2 | 2 | Alkaline Earth Metals |
| 3 | 13 | Boron Group |
| 4 | 14 | Carbon Group |
| 5 | 15 | Nitrogen Group |
| 6 | 16 | Chalcogens |
| 7 | 17 | Halogens |
| 8 | 2, 18 | Alkaline Earth Metals, Noble Gases |
| 9 | 3-12 | Transition Metals |
| 10 | 18 | Noble Gases |
| 11 | 17 | Halogens |
| 12 | 18 | Noble Gases |
| 13 | 14 | Carbon Group |
| 14 | 14-17 | Nonmetals |
| 15 | 17 | Halogens |
| 16 | 1 | Alkali Metals |
| 17 | 14 | Carbon Group |
| 18 | 18 | Noble Gases |
| 19 | 17 | Halogens |
| 20 | 2 | Alkaline Earth Metals |