QUESTION IMAGE
Question
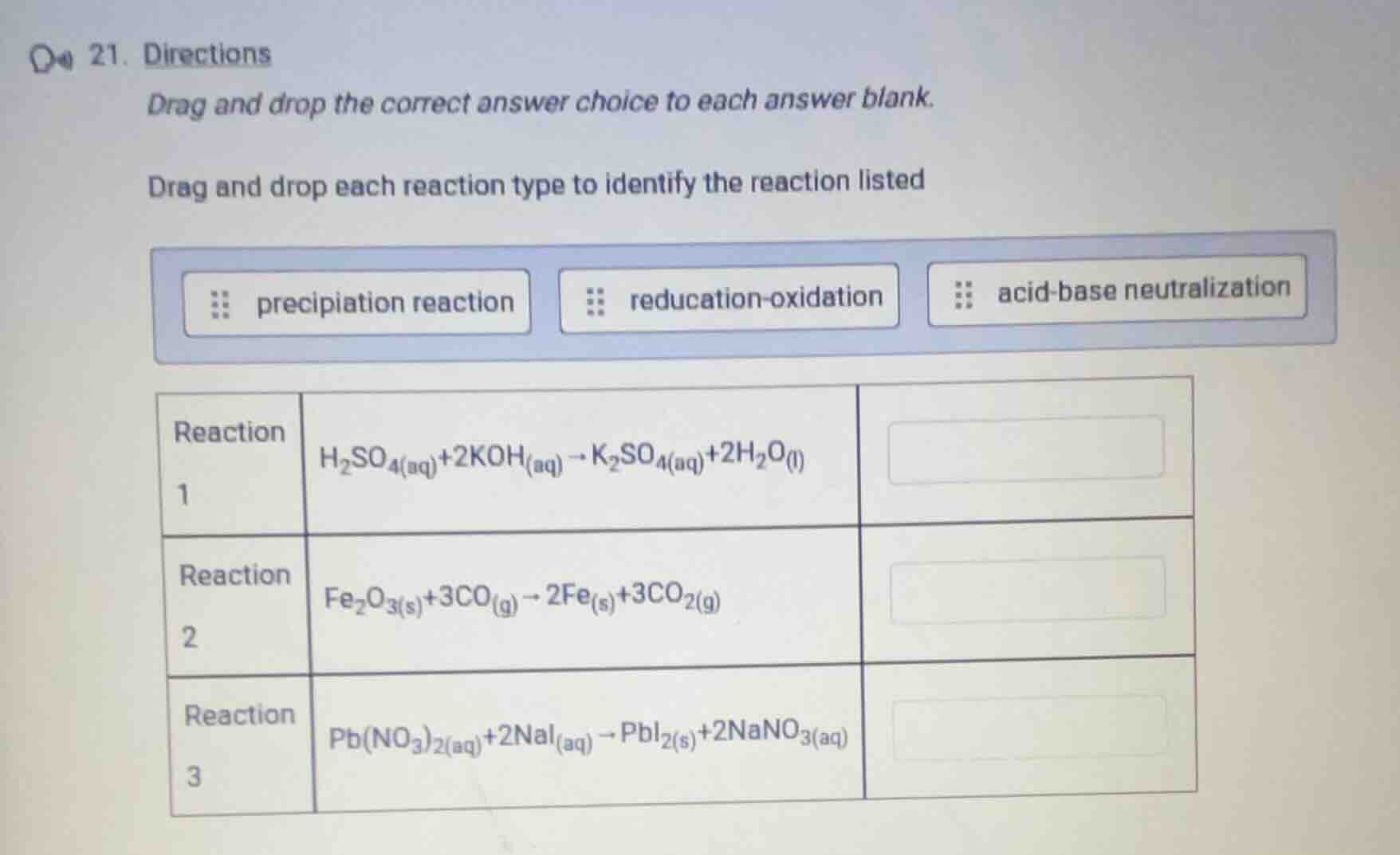
- directions
drag and drop the correct answer choice to each answer blank.
drag and drop each reaction type to identify the reaction listed
precipiation reaction reducation-oxidation acid-base neutralization
reaction 1: $ce{h_{2}so_{4}(aq) + 2koh(aq) -> k_{2}so_{4}(aq) + 2h_{2}o(l)}$
reaction 2: $ce{fe_{2}o_{3}(s) + 3co(g) -> 2fe(s) + 3co_{2}(g)}$
reaction 3: $ce{pb(no_{3})_{2}(aq) + 2nai(aq) -> pbi_{2}(s) + 2nano_{3}(aq)}$
Brief Explanations
- Reaction 1: Sulfuric acid ($\text{H}_2\text{SO}_4$), an acid, reacts with potassium hydroxide ($\text{KOH}$), a base, to produce a salt ($\text{K}_2\text{SO}_4$) and water, which matches the definition of an acid-base neutralization reaction.
- Reaction 2: Iron in $\text{Fe}_2\text{O}_3$ has an oxidation state of +3 and is reduced to 0 in $\text{Fe}$; carbon in $\text{CO}$ has an oxidation state of +2 and is oxidized to +4 in $\text{CO}_2$. The change in oxidation states confirms this is a redox (reduction-oxidation) reaction.
- Reaction 3: A solid precipitate ($\text{PbI}_2$) is formed from aqueous reactants, which is the defining characteristic of a precipitation reaction.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Reaction 1: acid-base neutralization
Reaction 2: reducation-oxidation
Reaction 3: precipiation reaction