QUESTION IMAGE
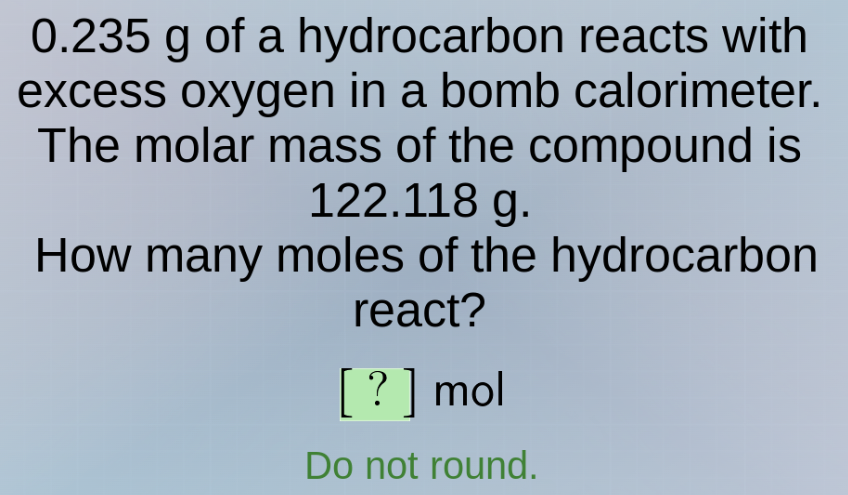
Question
0.235 g of a hydrocarbon reacts with excess oxygen in a bomb calorimeter. the molar mass of the compound is 122.118 g. how many moles of the hydrocarbon react? ? mol do not round.
Step1: Recall the formula for moles
The formula to calculate the number of moles (\(n\)) is \(n=\frac{m}{M}\), where \(m\) is the mass of the substance and \(M\) is the molar mass.
Step2: Substitute the given values
Here, the mass \(m = 0.235\space g\) and the molar mass \(M=122.118\space g/mol\). Substituting these values into the formula, we get \(n=\frac{0.235}{122.118}\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(\frac{0.235}{122.118}\) (or approximately \(0.001924\) but as per instruction not to round, we keep the fraction form or the decimal as is from the division)