QUESTION IMAGE
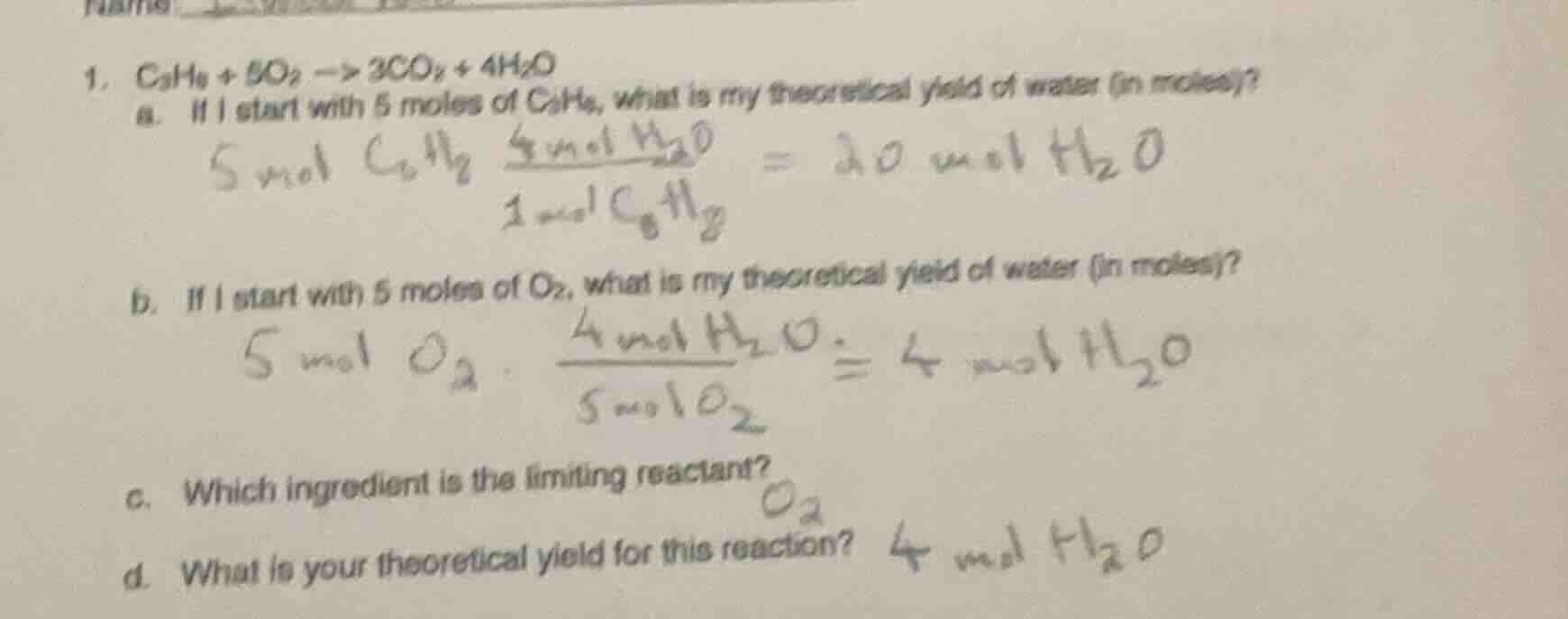
Question
- $c_3h_8 + 5o_2 \
ightarrow 3co_2 + 4h_2o$
a. if i start with 5 moles of $c_3h_8$, what is my theoretical yield of water (in moles)?
b. if i start with 5 moles of $o_2$, what is my theoretical yield of water (in moles)?
c. which ingredient is the limiting reactant?
d. what is your theoretical yield for this reaction?
Step1: Use mole ratio for $\text{C}_3\text{H}_8$
From the balanced equation $\text{C}_3\text{H}_8 + 5\text{O}_2
ightarrow 3\text{CO}_2 + 4\text{H}_2\text{O}$, the mole ratio of $\text{C}_3\text{H}_8$ to $\text{H}_2\text{O}$ is $1:4$.
$\text{Moles of } \text{H}_2\text{O} = 5\ \text{mol}\ \text{C}_3\text{H}_8 \times \frac{4\ \text{mol}\ \text{H}_2\text{O}}{1\ \text{mol}\ \text{C}_3\text{H}_8} = 20\ \text{mol}$
Step2: Use mole ratio for $\text{O}_2$
The mole ratio of $\text{O}_2$ to $\text{H}_2\text{O}$ is $5:4$.
$\text{Moles of } \text{H}_2\text{O} = 5\ \text{mol}\ \text{O}_2 \times \frac{4\ \text{mol}\ \text{H}_2\text{O}}{5\ \text{mol}\ \text{O}_2} = 4\ \text{mol}$
Step3: Identify limiting reactant
Compare yields from Step1 and Step2. The reactant producing less product is limiting. $\text{O}_2$ produces less $\text{H}_2\text{O}$.
Step4: Determine overall theoretical yield
The theoretical yield is the smaller product amount from limiting reactant.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a. $20$ moles
b. $4$ moles
c. $\text{O}_2$ (oxygen)
d. $4$ moles of $\text{H}_2\text{O}$