QUESTION IMAGE
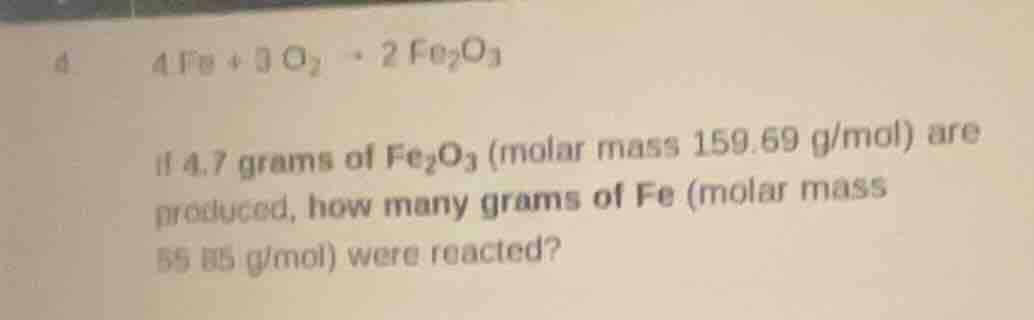
Question
- $4fe + 3o_2 \
ightarrow 2fe_2o_3$
if 4.7 grams of $fe_2o_3$ (molar mass 159.69 g/mol) are produced, how many grams of fe (molar mass 55.85 g/mol) were reacted?
Step1: Calculate moles of $\text{Fe}_2\text{O}_3$
$\text{Moles of Fe}_2\text{O}_3 = \frac{\text{Mass}}{\text{Molar Mass}} = \frac{4.7\ \text{g}}{159.69\ \text{g/mol}} \approx 0.0294\ \text{mol}$
Step2: Relate moles to $\text{Fe}$ via reaction
From $4\text{Fe} + 3\text{O}_2
ightarrow 2\text{Fe}_2\text{O}_3$, mole ratio $\text{Fe}:\text{Fe}_2\text{O}_3 = 4:2 = 2:1$
$\text{Moles of Fe} = 2 \times 0.0294\ \text{mol} = 0.0588\ \text{mol}$
Step3: Calculate mass of reacted $\text{Fe}$
$\text{Mass of Fe} = \text{Moles} \times \text{Molar Mass} = 0.0588\ \text{mol} \times 55.85\ \text{g/mol} \approx 3.28\ \text{g}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Approximately 3.3 grams (or 3.28 grams) of Fe were reacted.