QUESTION IMAGE
Question
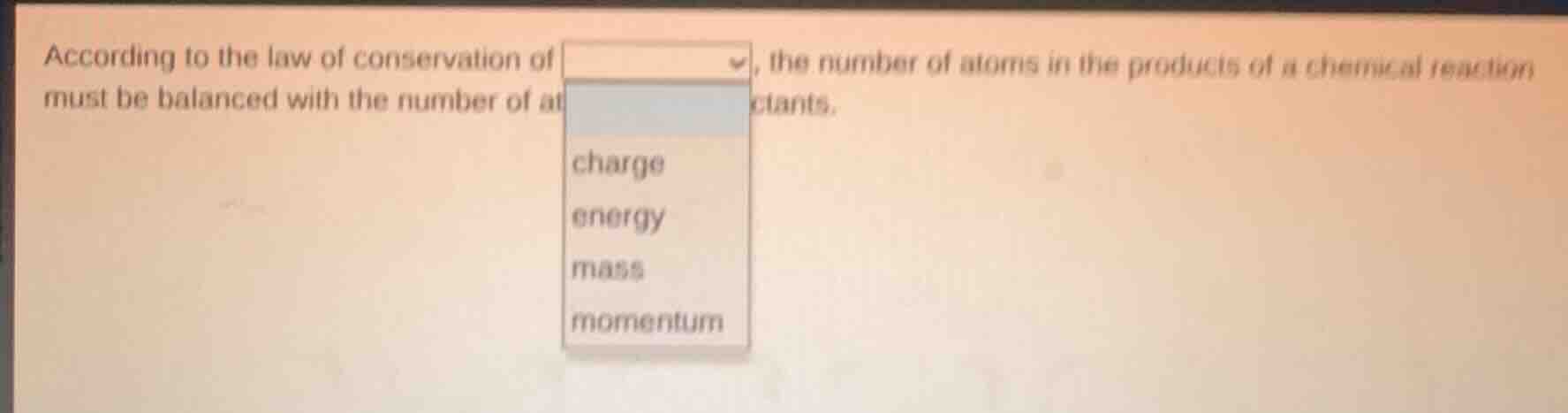
according to the law of conservation of, the number of atoms in the products of a chemical reaction must be balanced with the number of at ctants. charge energy mass momentum
The law of conservation of mass states that in a chemical reaction, matter is neither created nor destroyed. This means the total mass of reactants equals the total mass of products, and for this to hold, the number of atoms of each element in reactants must balance with those in products. Charge conservation is about electric charge, energy conservation is about energy (not directly about atom counts), and momentum conservation is about motion. So the correct term is mass.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
mass