QUESTION IMAGE
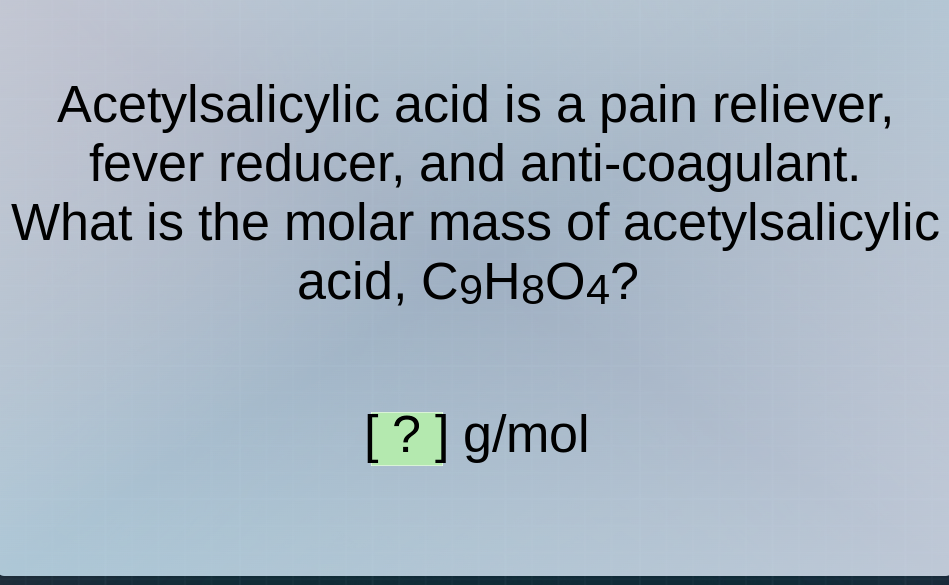
Question
acetylsalicylic acid is a pain reliever, fever reducer, and anti - coagulant. what is the molar mass of acetylsalicylic acid, $c_9h_8o_4$? ? g/mol
Step1: Find atomic masses
Atomic mass of C: \(12.01\) g/mol, H: \(1.008\) g/mol, O: \(16.00\) g/mol.
Step2: Calculate for each element
For C: \(9\times12.01 = 108.09\)
For H: \(8\times1.008 = 8.064\)
For O: \(4\times16.00 = 64.00\)
Step3: Sum the masses
Molar mass = \(108.09 + 8.064 + 64.00\)
\(= 180.154\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(180.15\) (or \(180.154\)) g/mol