QUESTION IMAGE
Question
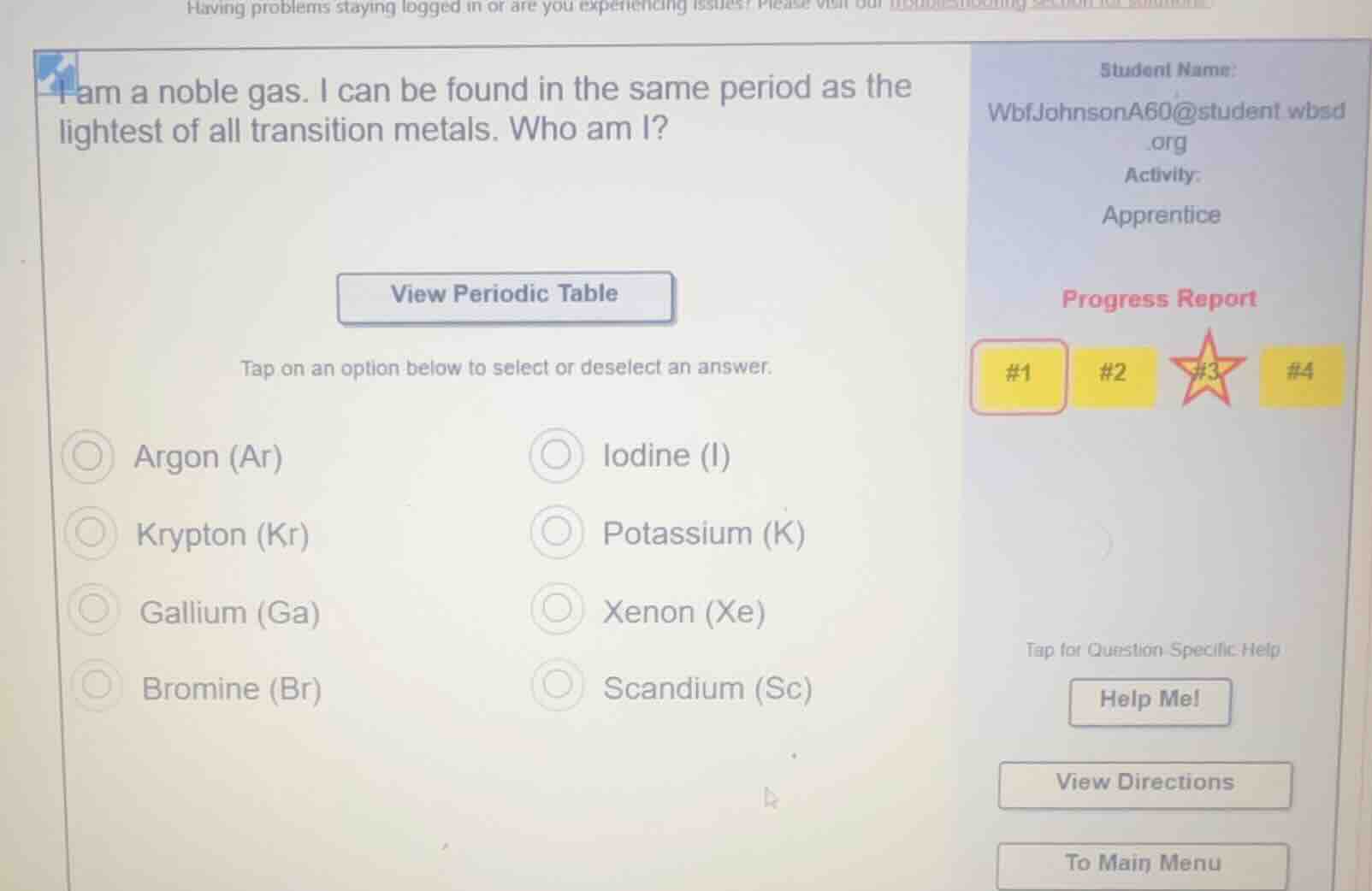
i am a noble gas. i can be found in the same period as the lightest of all transition metals. who am i?
view periodic table
tap on an option below to select or deselect an answer.
argon (ar)
iodine (i)
krypton (kr)
potassium (k)
gallium (ga)
xenon (xe)
bromine (br)
scandium (sc)
student name:
wbfjohnsona60@student.wbsd.org
activity:
apprentice
progress report
#1 #2 #3 #4
tap for question specific help
help me!
view directions
to main menu
Step1: Identify lightest transition metal
The lightest transition metal is Scandium (Sc), which is in Period 4 of the periodic table.
Step2: Locate Period 4 noble gas
Noble gases are in Group 18. The noble gas in Period 4 is Argon (Ar).
Step3: Eliminate non-noble gas options
Iodine, Potassium, Gallium, Bromine, Scandium are not noble gases, so they are eliminated. Krypton and Xenon are noble gases but in higher periods.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Argon (Ar)