QUESTION IMAGE
Question
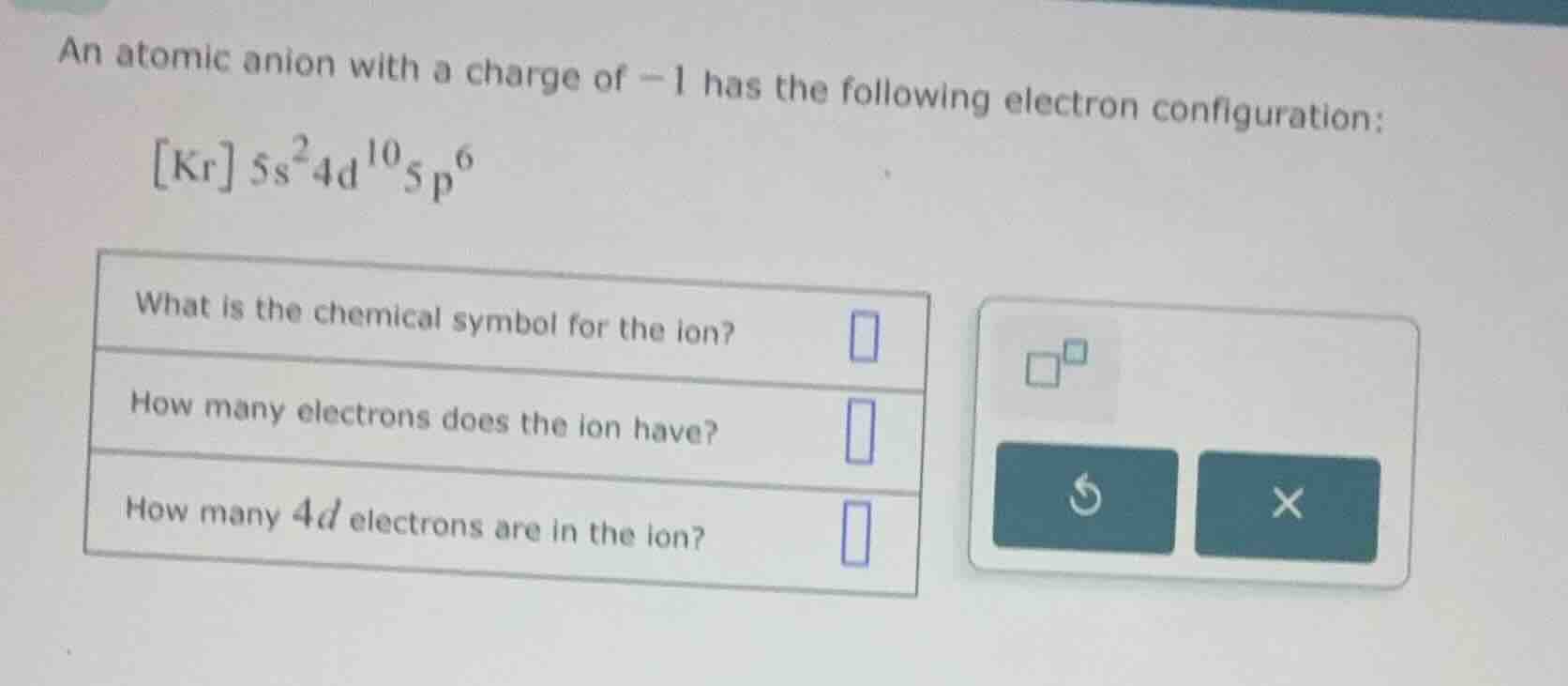
an atomic anion with a charge of $-1$ has the following electron configuration:
$kr 5s^24d^{10}5p^6$
what is the chemical symbol for the ion?
how many electrons does the ion have?
how many $4d$ electrons are in the ion?
Step1: Find Kr's electron count
Krypton (Kr) has 36 electrons.
Step2: Add valence electrons
Add electrons from $5s^2$, $4d^{10}$, $5p^6$: $36 + 2 + 10 + 6 = 54$
Step3: Identify neutral atom
The ion has a -1 charge, so neutral atom has $54 - 1 = 53$ electrons (iodine, I). Thus the ion is $\text{I}^-$.
Step4: Count 4d electrons
From configuration, $4d^{10}$ means 10 electrons.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{I}^-$
- 54
- 10