QUESTION IMAGE
Question
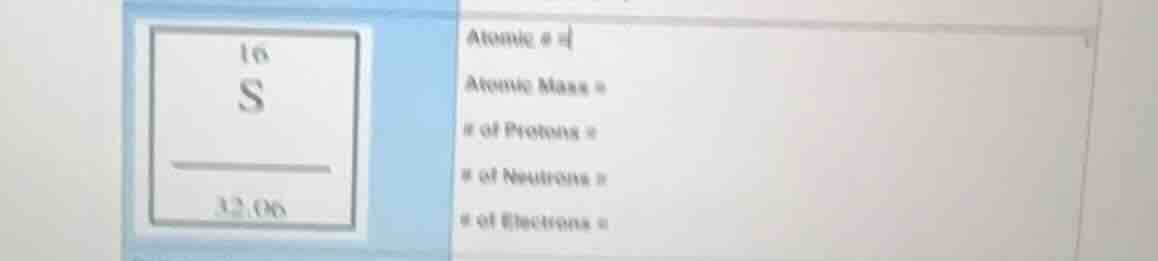
atomic # =
atomic mass =
of protons =
of neutrons =
of electrons =
Step 1: Determine Atomic Number
The atomic number (Z) is the number at the top of the element symbol in the periodic table box. For sulfur (S), the atomic number is 16. So, Atomic # = 16.
Step 2: Determine Atomic Mass
The atomic mass is the number at the bottom of the element symbol in the periodic table box. For sulfur, the atomic mass is 32.06. So, Atomic Mass = 32.06.
Step 3: Determine Number of Protons
In a neutral atom, the number of protons is equal to the atomic number. So, # of Protons = Atomic # = 16.
Step 4: Determine Number of Neutrons
The number of neutrons (n) is calculated by subtracting the atomic number from the atomic mass (rounded to the nearest whole number for simplicity, though sometimes the exact decimal is used). Atomic mass ≈ 32, so n = Atomic Mass (rounded) - Atomic Number = 32 - 16 = 16. So, # of Neutrons = 16 (or more precisely, 32.06 - 16 = 16.06, but usually rounded to 16).
Step 5: Determine Number of Electrons
In a neutral atom, the number of electrons is equal to the number of protons. So, # of Electrons = # of Protons = 16.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Atomic # = 16
Atomic Mass = 32.06