QUESTION IMAGE
Question
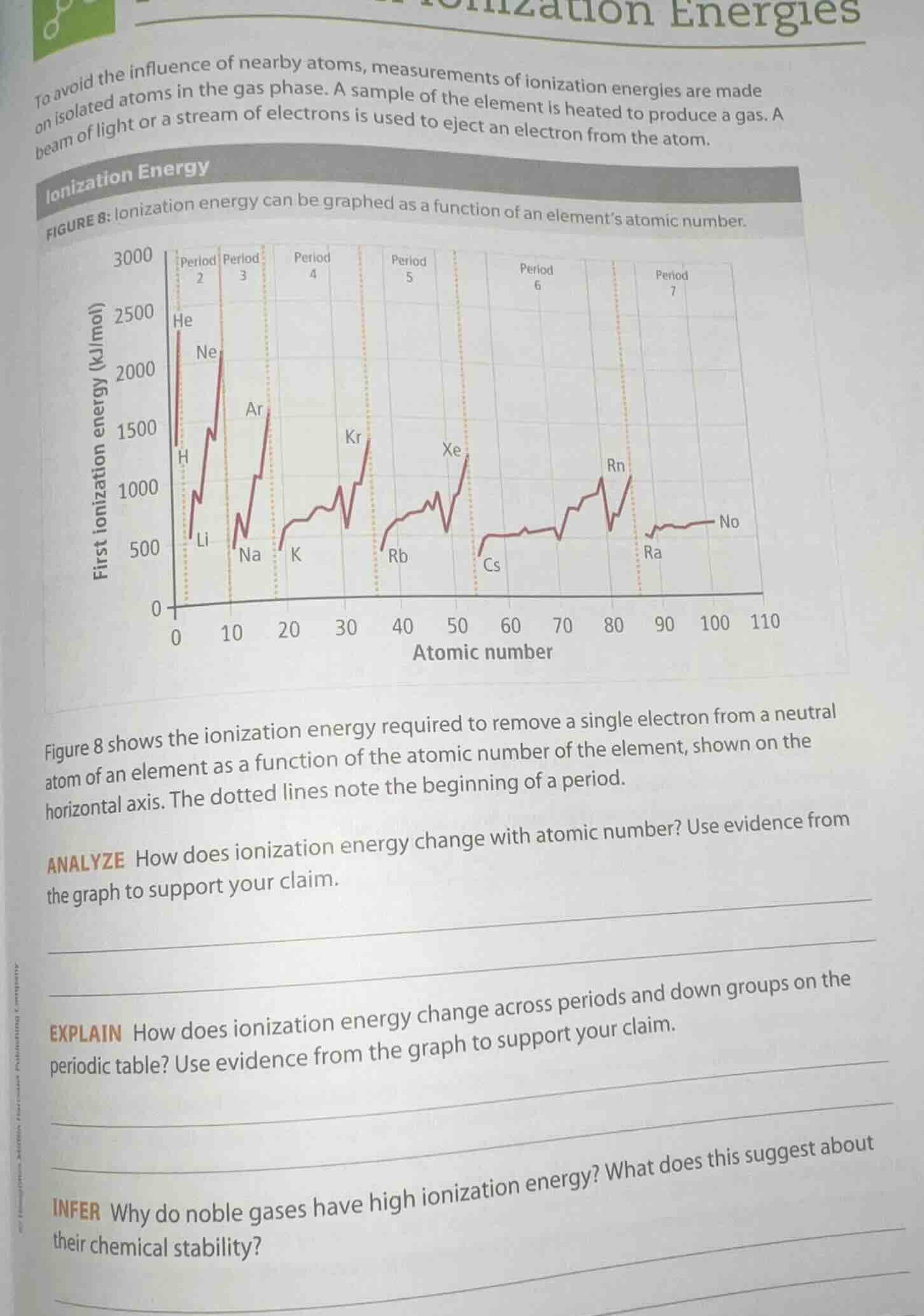
to avoid the influence of nearby atoms, measurements of ionization energies are made on isolated atoms in the gas phase. a sample of the element is heated to produce a gas. a beam of light or a stream of electrons is used to eject an electron from the atom.
ionization energy
figure 8: ionization energy can be graphed as a function of an element’s atomic number.
figure 8 shows the ionization energy required to remove a single electron from a neutral atom of an element as a function of the atomic number of the element, shown on the horizontal axis. the dotted lines note the beginning of a period.
analyze how does ionization energy change with atomic number? use evidence from the graph to support your claim.
explain how does ionization energy change across periods and down groups on the periodic table? use evidence from the graph to support your claim.
infer why do noble gases have high ionization energy? what does this suggest about their chemical stability?
ANALYZE
Ionization energy generally increases with atomic number in a periodic trend, with periodic peaks (at noble gases like He, Ne, Ar, Kr, Xe, Rn) and troughs (at alkali metals like Li, Na, K, Rb, Cs, Ra). For example, from Li (atomic number ~3) to Ne (atomic number 10), ionization energy rises; then drops at Na (atomic number 11) and rises again to Ar (atomic number 18), showing a repeating pattern.
Across a period (left to right, increasing atomic number), ionization energy increases. For example, in Period 2: Li (low) to Ne (high); Period 3: Na (low) to Ar (high). This is because as protons increase, the nucleus pulls electrons more strongly (same energy level, so shielding constant). Down a group (top to bottom, increasing atomic number), ionization energy decreases. For example, Li > Na > K > Rb > Cs (alkali metals) – electrons are in higher energy levels, farther from the nucleus, so easier to remove.
Noble gases (He, Ne, Ar, etc.) have high ionization energy because their electron configurations are stable (full valence shells, e.g., He: 1s², Ne: 2s²2p⁶). Removing an electron would disrupt this stability, requiring a lot of energy. This high ionization energy suggests they are chemically stable (unreactive) because they don’t easily lose electrons, and gaining electrons is also unfavorable (full shells), so they rarely form bonds.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Ionization energy shows a periodic trend with atomic number: it generally increases across a period (e.g., from Li to Ne, Na to Ar) and decreases down a group (e.g., Li > Na > K > Rb > Cs), with noble gases (He, Ne, Ar, etc.) having peak ionization energies and alkali metals (Li, Na, K, etc.) having troughs.