QUESTION IMAGE
Question
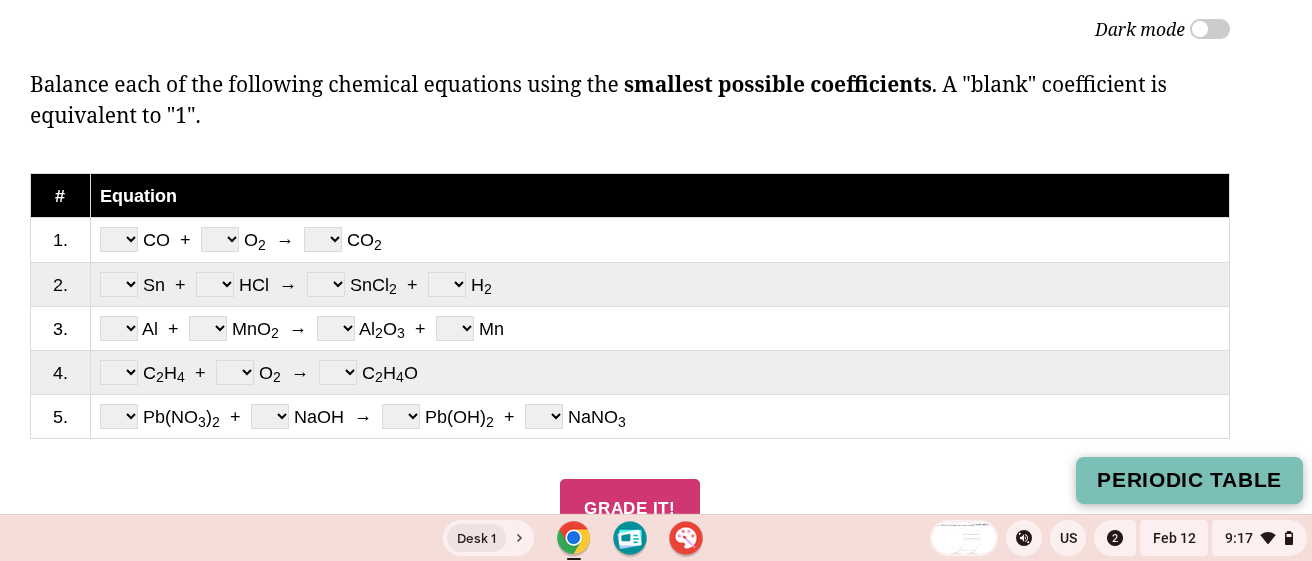
balance each of the following chemical equations using the smallest possible coefficients. a \blank\ coefficient is equivalent to \1\.
equation
- co + o₂ → co₂
- sn + hcl → sncl₂ + h₂
- al + mno₂ → al₂o₃ + mn
- c₂h₄ + o₂ → c₂h₄o
- pb(no₃)₂ + naoh → pb(oh)₂ + nano₃
Step1: Balance C, then O atoms
For $\text{CO} + \text{O}_2
ightarrow \text{CO}_2$:
- Balance C: 1 C on each side, so start with $1\text{CO} + \text{O}_2
ightarrow 1\text{CO}_2$
- Balance O: 1+2=3 O on left, 2 O on right. Multiply $\text{CO}$ and $\text{CO}_2$ by 2: $2\text{CO} + \text{O}_2
ightarrow 2\text{CO}_2$ (now 2+2=4 O on left, 4 O on right)
Step2: Balance Sn, Cl, then H atoms
For $\text{Sn} + \text{HCl}
ightarrow \text{SnCl}_2 + \text{H}_2$:
- Balance Sn: 1 Sn on each side, start with $1\text{Sn} + \text{HCl}
ightarrow 1\text{SnCl}_2 + \text{H}_2$
- Balance Cl: 2 Cl on right, so multiply $\text{HCl}$ by 2: $1\text{Sn} + 2\text{HCl}
ightarrow 1\text{SnCl}_2 + \text{H}_2$ (now 2 H on left, 2 H on right, balanced)
Step3: Balance Al, Mn, then O atoms
For $\text{Al} + \text{MnO}_2
ightarrow \text{Al}_2\text{O}_3 + \text{Mn}$:
- Balance Al: 2 Al on right, multiply $\text{Al}$ by 2: $2\text{Al} + \text{MnO}_2
ightarrow 1\text{Al}_2\text{O}_3 + \text{Mn}$
- Balance O: 2 O on left, 3 O on right. Use least common multiple 6: multiply $\text{MnO}_2$ by 3, $\text{Al}_2\text{O}_3$ by 2: $2\text{Al} + 3\text{MnO}_2
ightarrow 2\text{Al}_2\text{O}_3 + \text{Mn}$
- Rebalance Al: 4 Al on right, multiply $\text{Al}$ by 4: $4\text{Al} + 3\text{MnO}_2
ightarrow 2\text{Al}_2\text{O}_3 + \text{Mn}$
- Balance Mn: 3 Mn on left, multiply $\text{Mn}$ by 3: $4\text{Al} + 3\text{MnO}_2
ightarrow 2\text{Al}_2\text{O}_3 + 3\text{Mn}$
Step4: Balance C, H, then O atoms
For $\text{C}_2\text{H}_4 + \text{O}_2
ightarrow \text{C}_2\text{H}_4\text{O}$:
- Balance C and H: 2 C, 4 H on each side, start with $1\text{C}_2\text{H}_4 + \text{O}_2
ightarrow 1\text{C}_2\text{H}_4\text{O}$
- Balance O: 2 O on left, 1 O on right. Multiply $\text{C}_2\text{H}_4\text{O}$ by 2: $1\text{C}_2\text{H}_4 + \text{O}_2
ightarrow 2\text{C}_2\text{H}_4\text{O}$
- Rebalance C and H: 4 C, 8 H on right, multiply $\text{C}_2\text{H}_4$ by 2: $2\text{C}_2\text{H}_4 + \text{O}_2
ightarrow 2\text{C}_2\text{H}_4\text{O}$
- Balance O: 2 O on left, 2 O on right (balanced)
Step5: Balance Pb, NO₃, Na, then OH atoms
For $\text{Pb(NO}_3\text{)}_2 + \text{NaOH}
ightarrow \text{Pb(OH)}_2 + \text{NaNO}_3$:
- Balance Pb: 1 Pb on each side, start with $1\text{Pb(NO}_3\text{)}_2 + \text{NaOH}
ightarrow 1\text{Pb(OH)}_2 + \text{NaNO}_3$
- Balance $\text{NO}_3$: 2 $\text{NO}_3$ on left, multiply $\text{NaNO}_3$ by 2: $1\text{Pb(NO}_3\text{)}_2 + \text{NaOH}
ightarrow 1\text{Pb(OH)}_2 + 2\text{NaNO}_3$
- Balance Na: 2 Na on right, multiply $\text{NaOH}$ by 2: $1\text{Pb(NO}_3\text{)}_2 + 2\text{NaOH}
ightarrow 1\text{Pb(OH)}_2 + 2\text{NaNO}_3$ (now 2 OH on left, 2 OH on right, balanced)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 2, 1, 2
- 1, 2, 1, 1
- 4, 3, 2, 3
- 2, 1, 2
- 1, 2, 1, 2