QUESTION IMAGE
Question
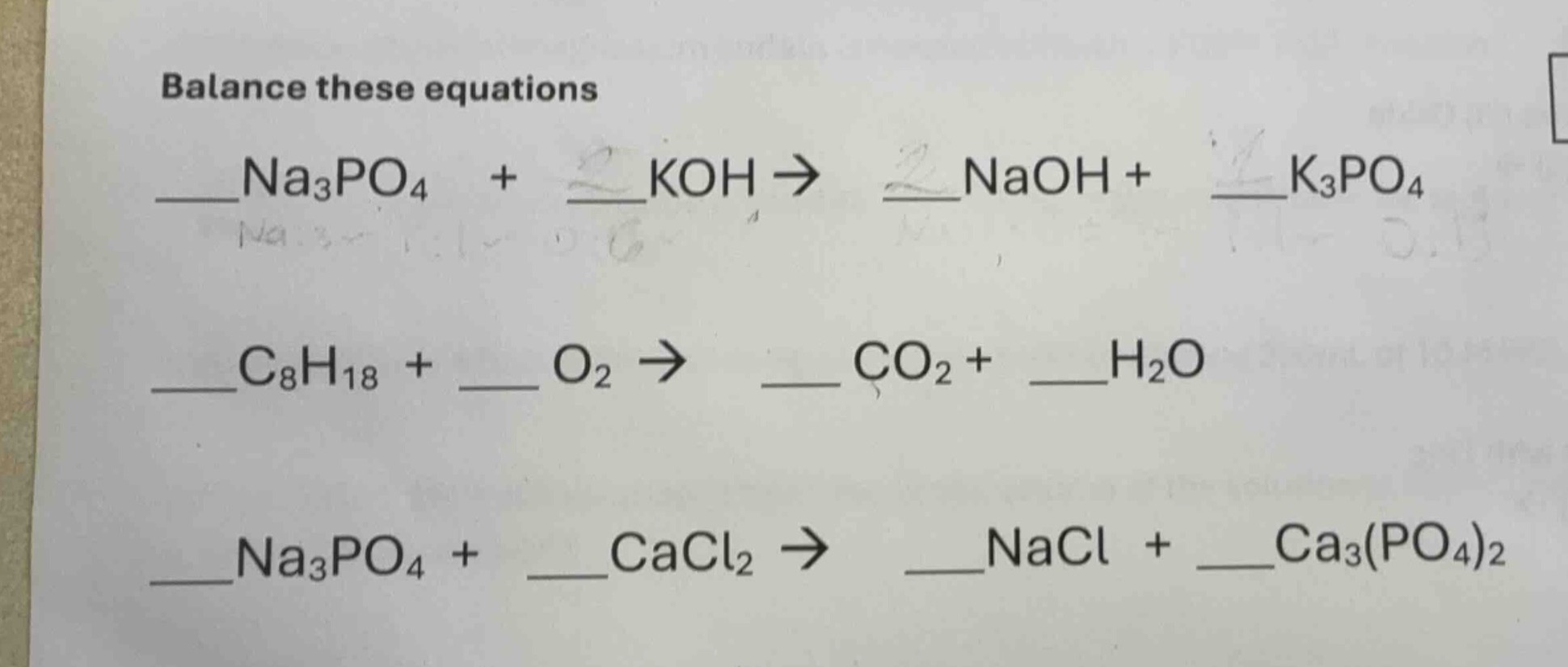
balance these equations
__na₃po₄ + koh → naoh + __k₃po₄
__c₈h₁₈ + o₂ → co₂ + __h₂o
__na₃po₄ + cacl₂ → nacl + __ca₃(po₄)₂
Step1: Balance 1st equation (Na, K)
- Count Na: Left has 3 in $\text{Na}_3\text{PO}_4$, right has 1 in $\text{NaOH}$. Assign 3 to $\text{NaOH}$.
- Count K: Right has 3 in $\text{K}_3\text{PO}_4$, left has 1 in $\text{KOH}$. Assign 3 to $\text{KOH}$.
- Verify all atoms: $\text{Na}_3\text{PO}_4 + 3\text{KOH}
ightarrow 3\text{NaOH} + \text{K}_3\text{PO}_4$ (all atoms balanced)
Step2: Balance 2nd equation (C, H, O)
- Count C: Left has 8 in $\text{C}_8\text{H}_{18}$, assign 8 to $\text{CO}_2$.
- Count H: Left has 18 in $\text{C}_8\text{H}_{18}$, right has 2 in $\text{H}_2\text{O}$. Assign 9 to $\text{H}_2\text{O}$.
- Count O: Right has $8\times2 + 9\times1=25$, left has 2 in $\text{O}_2$. Assign $\frac{25}{2}$ to $\text{O}_2$, then multiply all coefficients by 2 to eliminate fractions:
$2\text{C}_8\text{H}_{18} + 25\text{O}_2
ightarrow 16\text{CO}_2 + 18\text{H}_2\text{O}$ (all atoms balanced)
Step3: Balance 3rd equation (Ca, PO₄, Na, Cl)
- Count Ca: Right has 3 in $\text{Ca}_3(\text{PO}_4)_2$, left has 1 in $\text{CaCl}_2$. Assign 3 to $\text{CaCl}_2$.
- Count $\text{PO}_4$: Right has 2 in $\text{Ca}_3(\text{PO}_4)_2$, left has 1 in $\text{Na}_3\text{PO}_4$. Assign 2 to $\text{Na}_3\text{PO}_4$.
- Count Na: Left has $2\times3=6$ in $\text{Na}_3\text{PO}_4$, right has 1 in $\text{NaCl}$. Assign 6 to $\text{NaCl}$.
- Verify Cl: Left has $3\times2=6$ in $\text{CaCl}_2$, right has 6 in $\text{NaCl}$. Balanced:
$2\text{Na}_3\text{PO}_4 + 3\text{CaCl}_2
ightarrow 6\text{NaCl} + \text{Ca}_3(\text{PO}_4)_2$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\boldsymbol{\text{Na}_3\text{PO}_4 + 3\text{KOH}
ightarrow 3\text{NaOH} + \text{K}_3\text{PO}_4}$ (coefficients: 1, 3, 3, 1)
- $\boldsymbol{2\text{C}_8\text{H}_{18} + 25\text{O}_2
ightarrow 16\text{CO}_2 + 18\text{H}_2\text{O}}$ (coefficients: 2, 25, 16, 18)
- $\boldsymbol{2\text{Na}_3\text{PO}_4 + 3\text{CaCl}_2
ightarrow 6\text{NaCl} + \text{Ca}_3(\text{PO}_4)_2}$ (coefficients: 2, 3, 6, 1)