QUESTION IMAGE
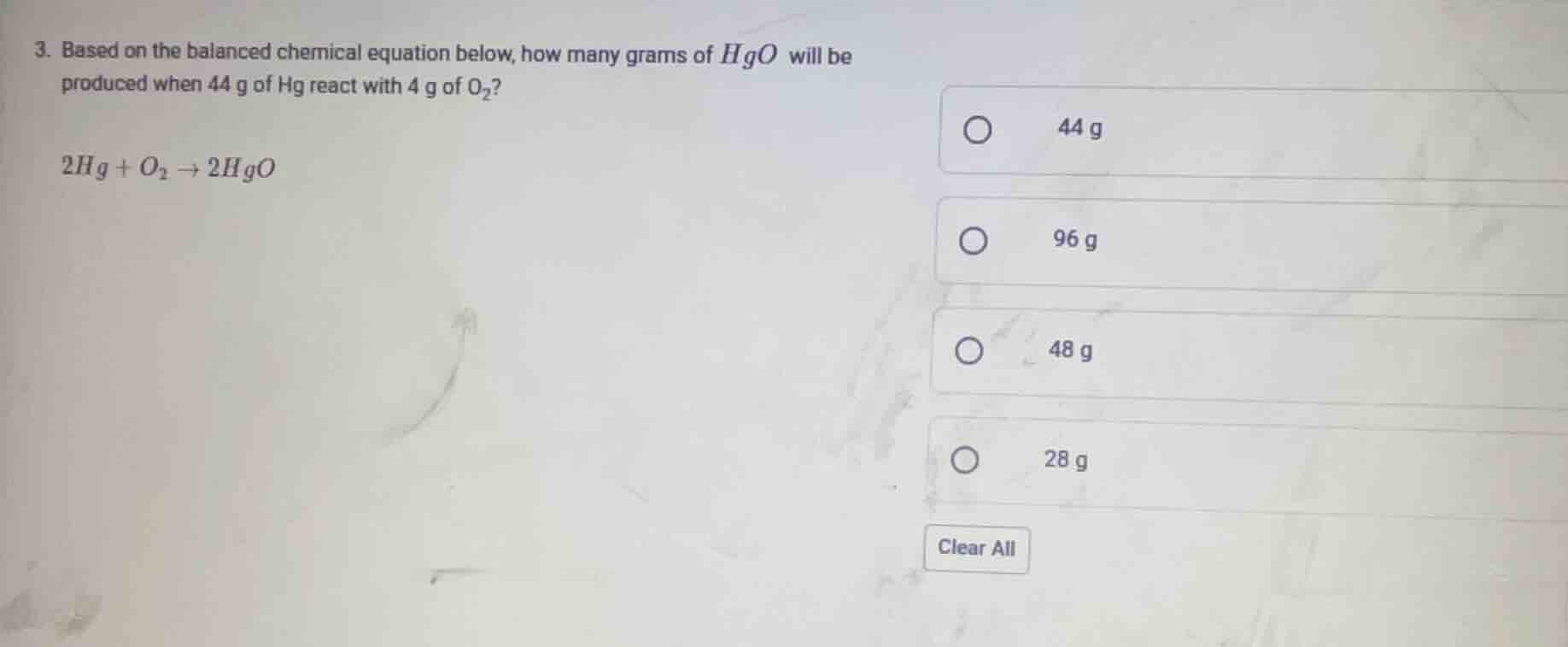
Question
- based on the balanced chemical equation below, how many grams of $hgo$ will be produced when 44 g of hg react with 4 g of $o_2$?
$2hg + o_2 \
ightarrow 2hgo$
44 g
96 g
48 g
28 g
clear all
Step1: Calculate molar masses
Molar mass of $Hg$: $200.6\ \text{g/mol}$, molar mass of $O_2$: $32\ \text{g/mol}$, molar mass of $HgO$: $200.6 + 16 = 216.6\ \text{g/mol}$
Step2: Find moles of reactants
Moles of $Hg$: $\frac{44\ \text{g}}{200.6\ \text{g/mol}} \approx 0.219\ \text{mol}$
Moles of $O_2$: $\frac{4\ \text{g}}{32\ \text{g/mol}} = 0.125\ \text{mol}$
Step3: Identify limiting reactant
From $2Hg + O_2
ightarrow 2HgO$, 2 mol $Hg$ reacts with 1 mol $O_2$.
Required $O_2$ for $0.219\ \text{mol}\ Hg$: $\frac{0.219}{2} = 0.1095\ \text{mol}$.
We have excess $O_2$, so $Hg$ is limiting.
Step4: Calculate moles of $HgO$
Moles of $HgO$ = moles of limiting $Hg$ = $0.219\ \text{mol}$
Step5: Calculate mass of $HgO$
Mass = $0.219\ \text{mol} \times 216.6\ \text{g/mol} \approx 47.4\ \text{g} \approx 48\ \text{g}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
48 g