QUESTION IMAGE
Question
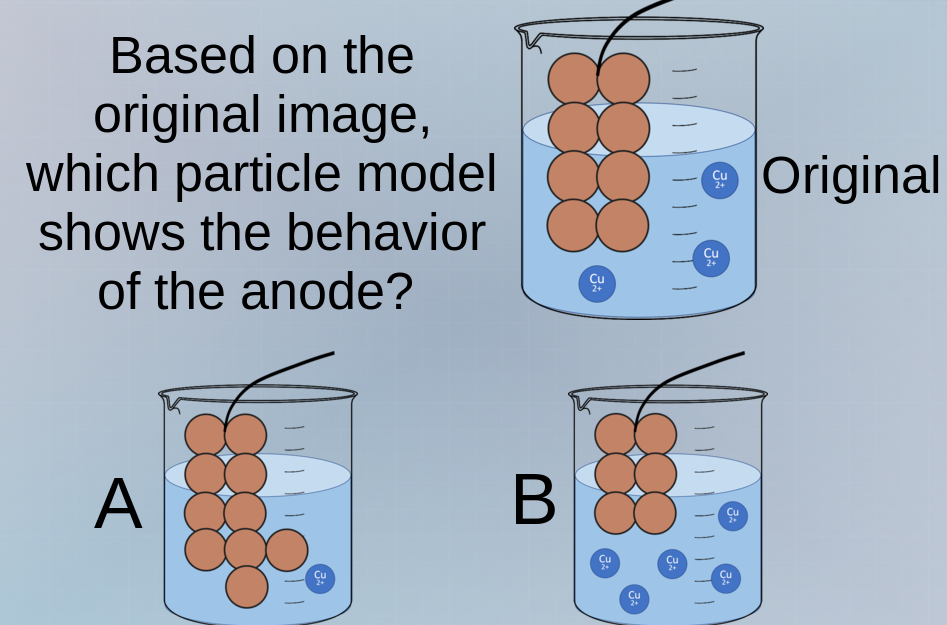
based on the original image, which particle model shows the behavior of the anode? a b original
In an electrochemical cell, the anode is where oxidation occurs (loss of electrons). For a copper anode (assuming the original has a copper electrode), the anode reaction is \( \text{Cu}(s)
ightarrow \text{Cu}^{2+}(aq) + 2e^- \). This means solid copper atoms from the anode dissolve into the solution as \( \text{Cu}^{2+} \) ions, so the number of solid copper particles (the brown spheres) should decrease, and the number of \( \text{Cu}^{2+} \) ions in the solution should increase.
- In the original image, there are 8 brown spheres (anode) and some \( \text{Cu}^{2+} \) ions.
- In option A: The number of brown spheres (solid Cu) has increased (now 9), which is incorrect because the anode should lose mass (dissolve), not gain.
- In option B: The number of brown spheres (solid Cu) has decreased (now 6), and the number of \( \text{Cu}^{2+} \) ions in the solution has increased (more blue \( \text{Cu}^{2+} \) ions), which matches the anode oxidation (dissolution of Cu into \( \text{Cu}^{2+} \)).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. The particle model with 6 brown spheres (solid Cu) and more \( \text{Cu}^{2+} \) ions in the solution.