QUESTION IMAGE
Question
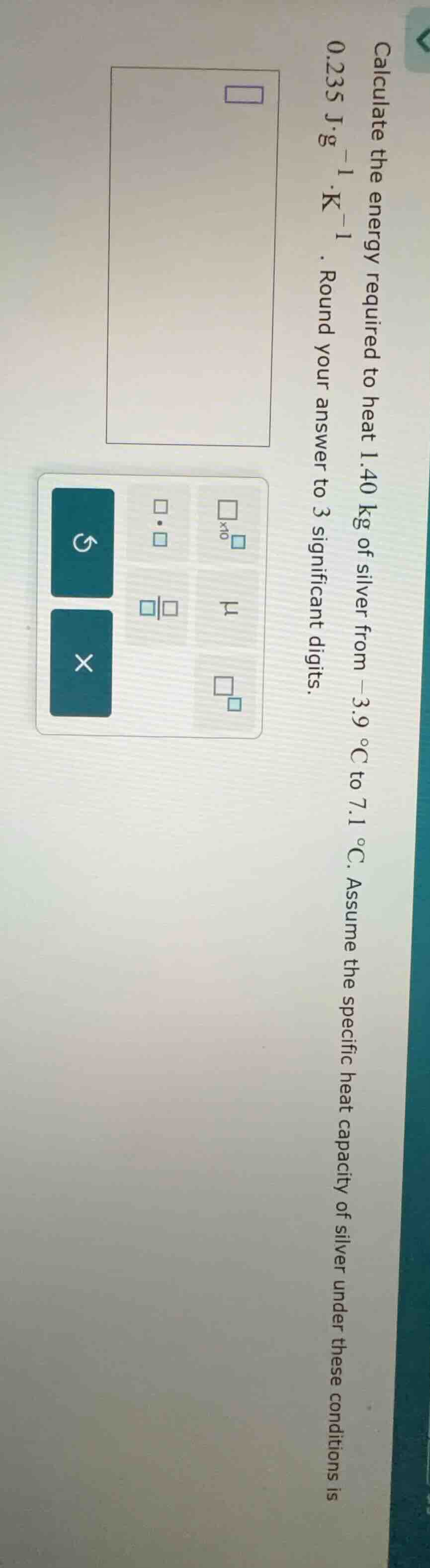
calculate the energy required to heat 1.40 kg of silver from -3.9 °c to 7.1 °c. assume the specific heat capacity of silver under these conditions is 0.235 j·g⁻¹·k⁻¹. round your answer to 3 significant digits.
Step1: Convert mass to grams
The mass of silver is \( 1.40 \, \text{kg} \). Since \( 1 \, \text{kg} = 1000 \, \text{g} \), we convert the mass:
\( m = 1.40 \, \text{kg} \times 1000 \, \text{g/kg} = 1400 \, \text{g} \)
Step2: Calculate temperature change
The initial temperature \( T_1 = -3.9 \, ^\circ\text{C} \) and the final temperature \( T_2 = 7.1 \, ^\circ\text{C} \). The temperature change \( \Delta T \) is:
\( \Delta T = T_2 - T_1 = 7.1 \, ^\circ\text{C} - (-3.9 \, ^\circ\text{C}) = 11.0 \, ^\circ\text{C} \) (Note: A change in Celsius is the same as a change in Kelvin, so \( \Delta T \) in K is also \( 11.0 \, \text{K} \))
Step3: Use the heat formula \( q = mc\Delta T \)
The specific heat capacity \( c = 0.235 \, \text{J} \cdot \text{g}^{-1} \cdot \text{K}^{-1} \), mass \( m = 1400 \, \text{g} \), and \( \Delta T = 11.0 \, \text{K} \). Plugging into the formula:
\( q = 1400 \, \text{g} \times 0.235 \, \text{J} \cdot \text{g}^{-1} \cdot \text{K}^{-1} \times 11.0 \, \text{K} \)
First, calculate \( 1400 \times 0.235 = 329 \)
Then, \( 329 \times 11.0 = 3619 \, \text{J} \)
Step4: Round to 3 significant digits
\( 3619 \, \text{J} \) rounded to 3 significant digits is \( 3620 \, \text{J} \) or \( 3.62 \times 10^3 \, \text{J} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 3.62 \times 10^3 \, \text{J} \) (or \( 3620 \, \text{J} \))