QUESTION IMAGE
Question
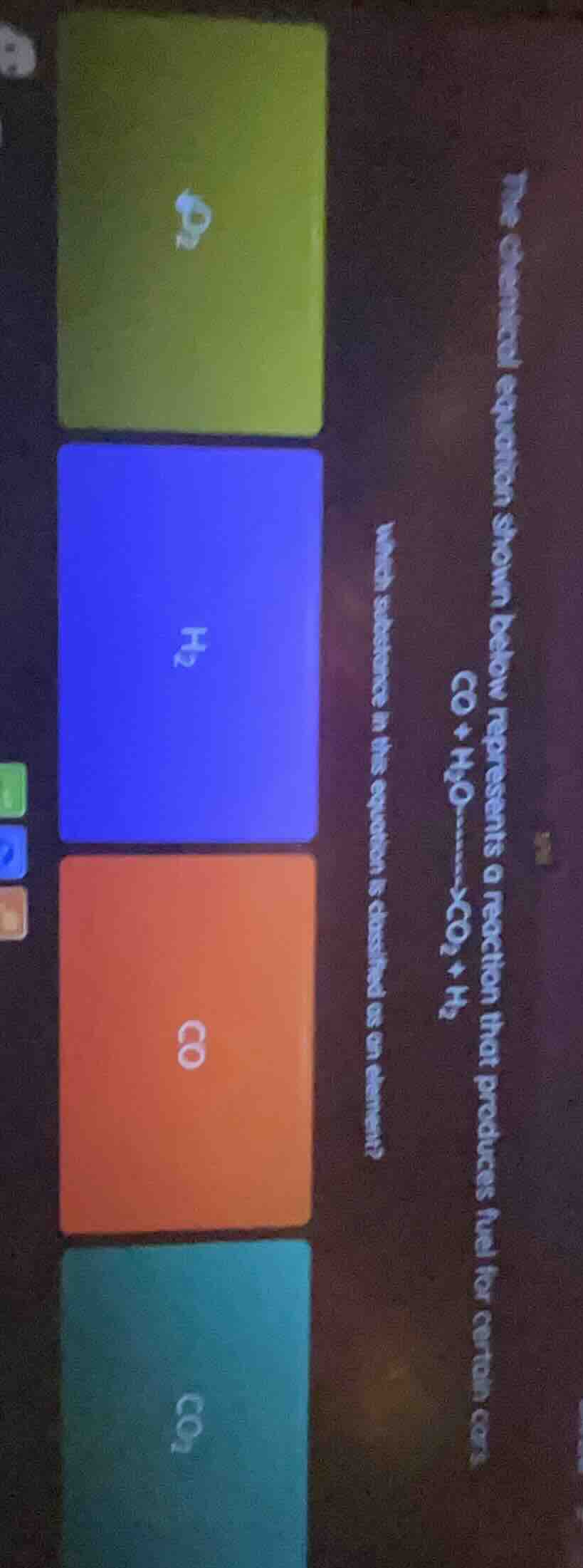
the chemical equation shown below represents a reaction that produces fuel for certain cars?\
$\ce{co + h_{2}o -> xco_{2} + h_{2}}$\
which substance in this equation is classified as an element?\
(options: $\ce{co_{2}}$, $\ce{h_{2}}$, $\ce{co}$, $\ce{co_{2}}$ (with corresponding colored boxes))
An element is a pure substance made of only one type of atom. Let's analyze each substance:
- \( \text{CO} \) (carbon monoxide) is a compound made of carbon and oxygen atoms.
- \( \text{CO}_2 \) (carbon dioxide) is a compound made of carbon and oxygen atoms.
- \( \text{H}_2 \) (hydrogen gas) is made of only hydrogen atoms, so it is an element.
- The other option (not fully clear but from the choices, \( \text{H}_2 \) fits the definition of an element as it consists of only one type of atom (hydrogen), while \( \text{CO} \) and \( \text{CO}_2 \) are compounds with multiple types of atoms.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The substance classified as an element is \( \text{H}_2 \) (Hydrogen gas). If we consider the options as presented (assuming the choices are \( \text{CO}_2 \), \( \text{H}_2 \), \( \text{CO} \), \( \text{CO}_2 \) etc.), the correct one is the one with \( \text{H}_2 \) (e.g., if options are labeled, say B. \( \text{H}_2 \), then B. \( \text{H}_2 \)).