QUESTION IMAGE
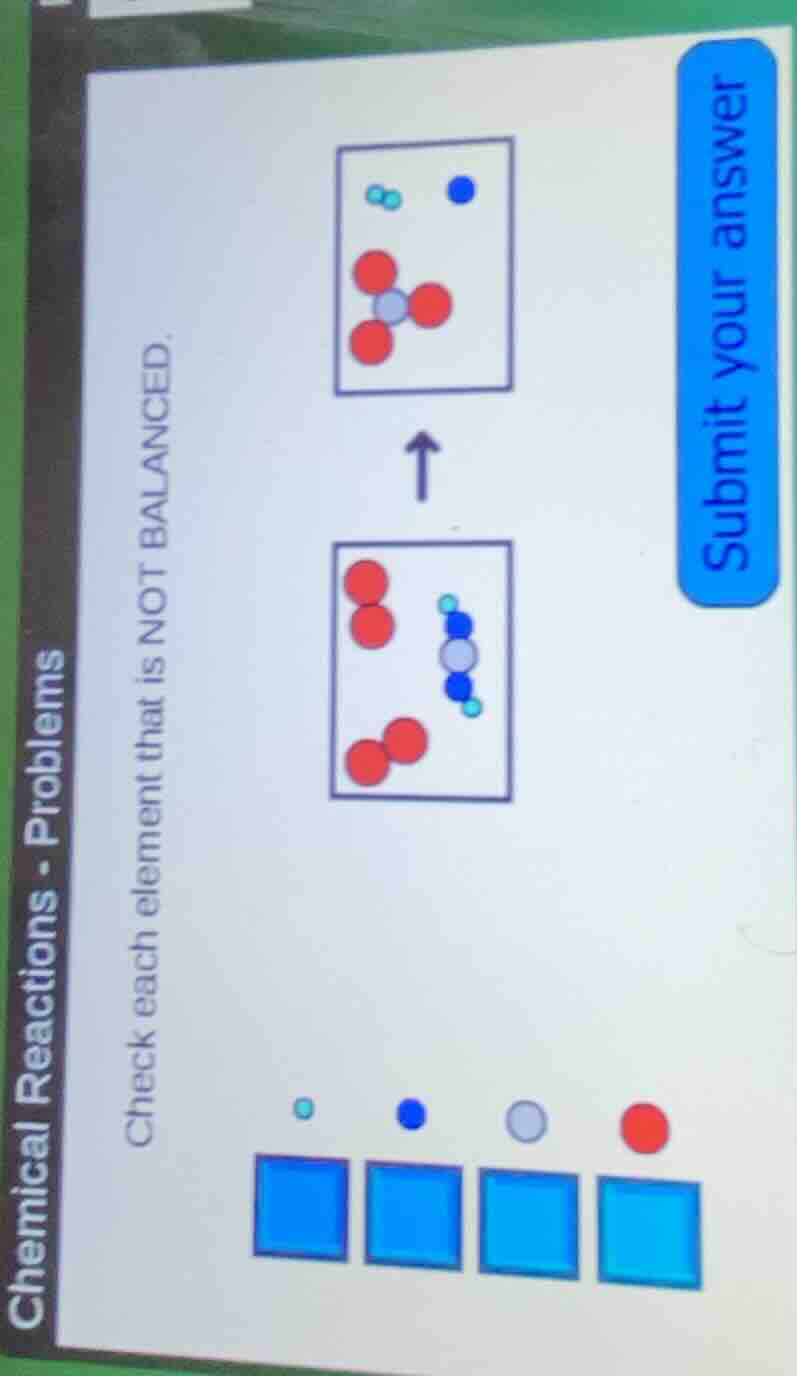
Question
chemical reactions - problems
check each element that is not balanced.
submit your answer
Step1: Count atoms of each color (element) in reactants (bottom box)
- Light blue (let's say H): In the molecule, there are 2 (one on left, one on right). Plus the other? Wait, the reactant has one molecule with 2 light blue? Wait, no, the reactant (bottom) has a molecule with 2 blue (dark blue, N?), 1 gray (C?), 2 light blue (H). Then two red diatoms (O₂): each O₂ has 2 red, so two O₂ have 4 red. Gray: 1. Dark blue: 2. Light blue: 2.
Step2: Count atoms in products (top box)
- Light blue: The small molecule has 2, and the other? Wait, top box: small light blue molecule (2), dark blue: 1, gray: 1, red: 3 (from the gray - red molecule: 3 red, and is there another red? No. So red in products: 3. Light blue: 2 (from small molecule) + 0? Wait, the gray - red molecule has no light blue. Dark blue: 1. Gray: 1.
Step3: Compare each element
- Red (O): Reactants have 4 (2 O₂, each with 2), products have 3 (from the gray - red molecule: 3 red). Not balanced.
- Dark blue (N): Reactants have 2, products have 1. Not balanced.
- Light blue (H): Reactants have 2, products have 2 (small molecule). Balanced.
- Gray (C): Reactants have 1, products have 1. Balanced.
Wait, let's re - count:
Reactants (bottom box):
- Red (O): Two O₂ molecules. Each O₂ has 2 O atoms. So 2×2 = 4 O atoms.
- Dark blue (N): The molecule has 2 N atoms.
- Light blue (H): The molecule has 2 H atoms.
- Gray (C): The molecule has 1 C atom.
Products (top box):
- Red (O): The C - O₃ molecule has 3 O atoms.
- Dark blue (N): 1 N atom.
- Light blue (H): The H₂ molecule has 2 H atoms.
- Gray (C): 1 C atom.
So for N: Reactants have 2, products have 1. Not balanced.
For O: Reactants have 4, products have 3. Not balanced.
But let's check the boxes:
Wait, the bottom box (reactants):
- Red: two pairs (so 2×2 = 4)
- Dark blue: two in a molecule with gray and light blue
- Light blue: two in that molecule
- Gray: one in that molecule
Top box (products):
- Red: three in the gray - red molecule
- Dark blue: one single
- Light blue: two in a small molecule
- Gray: one in the gray - red molecule
So the elements (colors) that are not balanced: Red (O) and Dark blue (N). But let's see the options (the boxes to check: the four boxes corresponding to light blue, dark blue, gray, red).
Wait, the question is to check each element that is NOT BALANCED.
Let's list each:
- Light blue (H): Reactants: 2, Products: 2 (small H₂ molecule). Balanced. So don't check.
- Dark blue (N): Reactants: 2, Products: 1. Not balanced. Check.
- Gray (C): Reactants: 1, Products: 1. Balanced. Don't check.
- Red (O): Reactants: 4, Products: 3. Not balanced. Check.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The elements (colors) that are not balanced are the dark blue (N) and red (O) and also the dark blue (N) and red (O) are not balanced. But looking at the boxes (the four squares for light blue, dark blue, gray, red), we check the dark blue (N) and red (O) squares. Wait, maybe the intended answer is dark blue (N) and red (O), but let's see the count again.
Wait, maybe the dark blue is N, light blue is H, gray is C, red is O.
Reactants:
- C: 1
- H: 2
- N: 2
- O: 4 (2 O₂)
Products:
- C: 1
- H: 2 (H₂)
- N: 1 (single N)
- O: 3 (CO₃? Wait, the gray - red molecule: C with 3 O? So CO₃? Then O is 3)
So N: 2→1 (not balanced), O: 4→3 (not balanced), H: 2→2 (balanced), C: 1→1 (balanced).
So the elements (colors) that are not balanced are dark blue (N) and red (O). So we should check the boxes for dark blue and red. But maybe the question is to check each element that is not balanced. So the answer is that the dark blue (second box) and red (fourth box) elements are not balanced.