QUESTION IMAGE
Question
chemistry resources
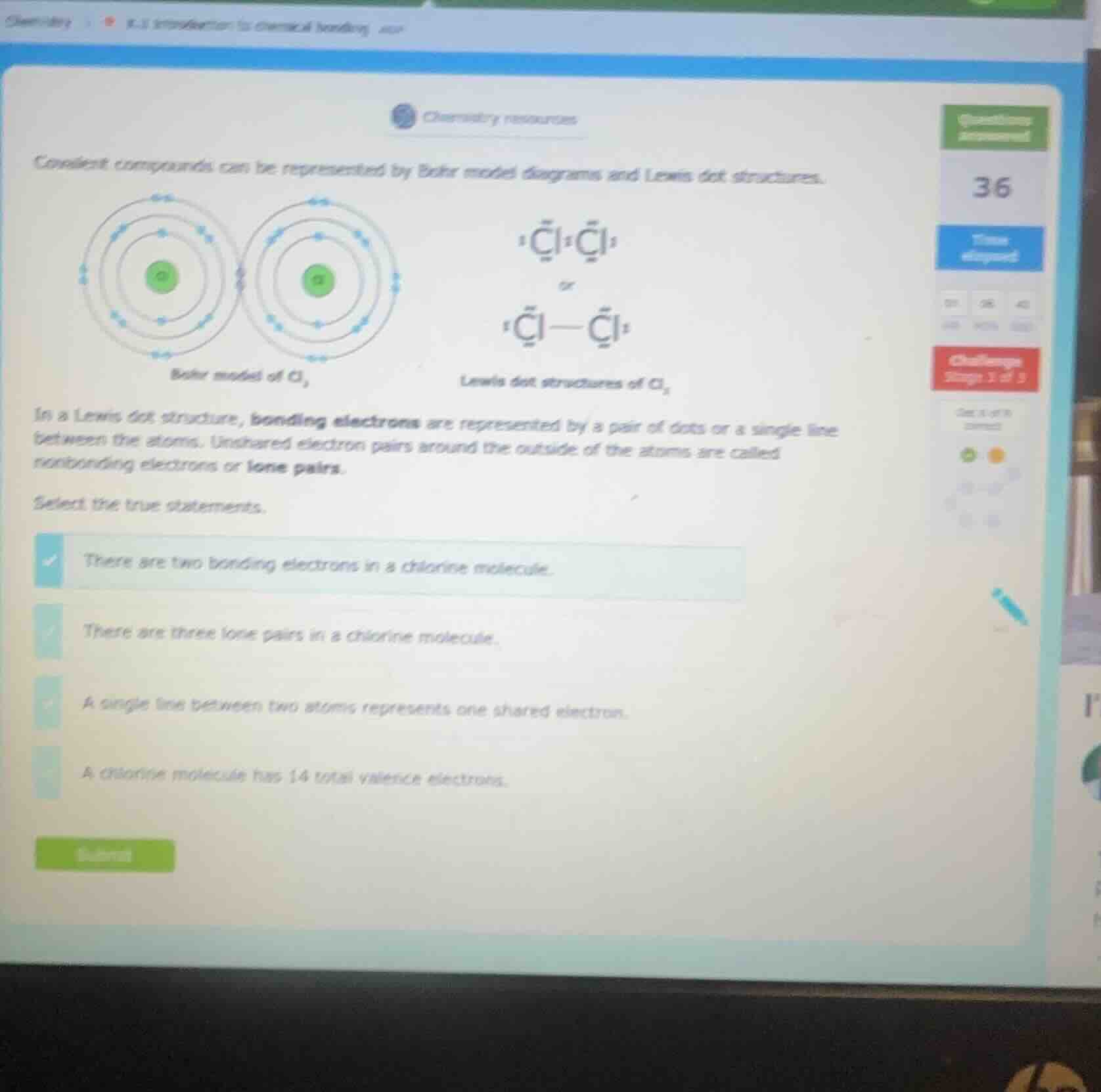
covalent compounds can be represented by bohr model diagrams and lewis dot structures.
bohr model of cl₂
lewis dot structures of cl₂
in a lewis dot structure, bonding electrons are represented by a pair of dots or a single line between the atoms. unshared electron pairs around the outside of the atoms are called nonbonding electrons or lone pairs.
select the true statements.
- there are two bonding electrons in a chlorine molecule.
- there are three lone pairs in a chlorine molecule.
- a single line between two atoms represents one shared electron.
- a chlorine molecule has 14 total valence electrons.
Brief Explanations
- For the first statement: The Cl₂ molecule has a single covalent bond, which consists of 2 shared (bonding) electrons. This is true.
- For the second statement: Each Cl atom in Cl₂ has 3 lone pairs, so the total lone pairs in the molecule are 6, not 3. This is false.
- For the third statement: A single line in a Lewis structure represents a pair of shared electrons (2 total), not 1. This is false.
- For the fourth statement: A single Cl atom has 7 valence electrons, so two Cl atoms have \(7 \times 2 = 14\) total valence electrons. This is true.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- There are two bonding electrons in a chlorine molecule.
- A chlorine molecule has 14 total valence electrons.