QUESTION IMAGE
Question
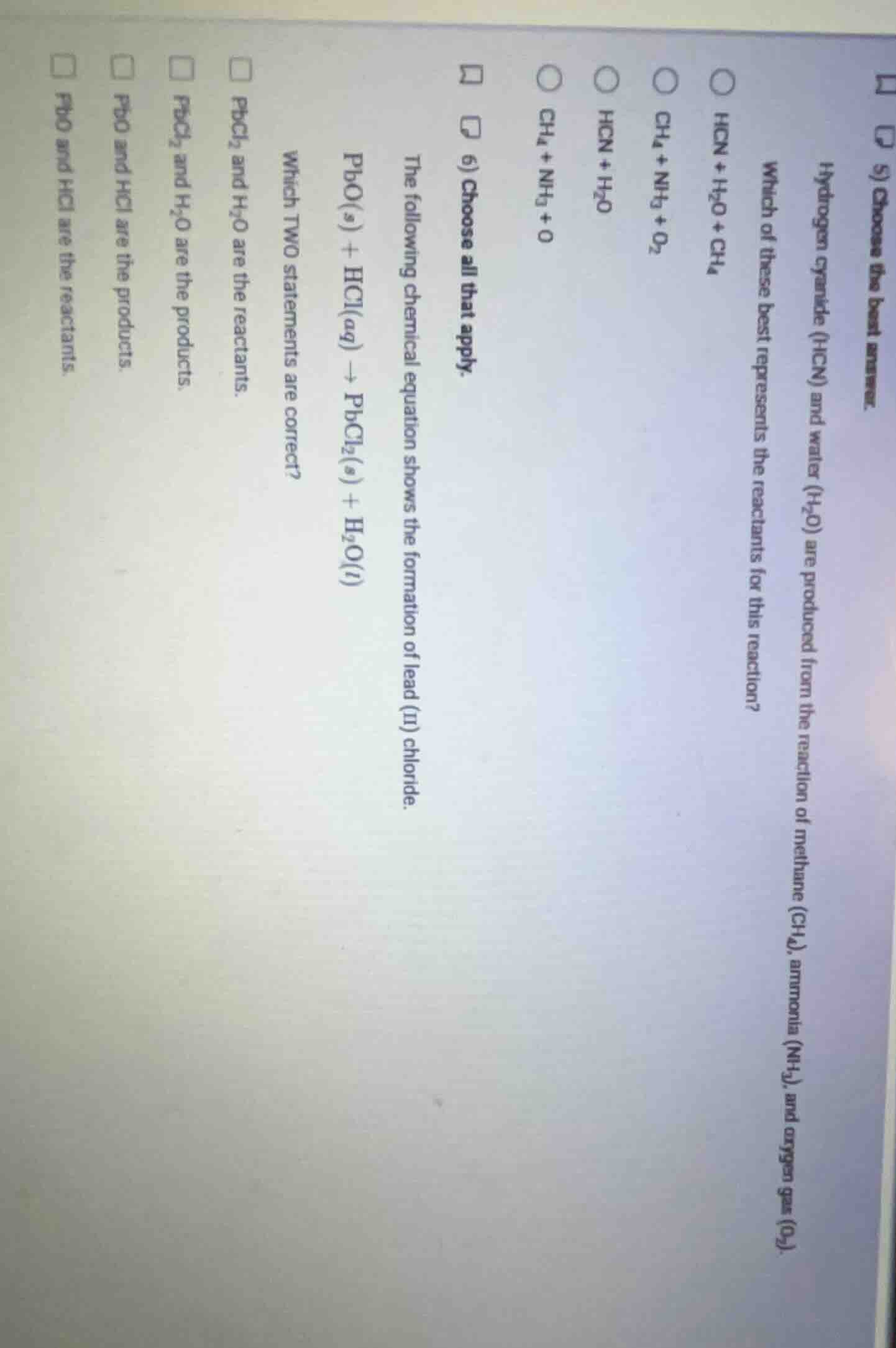
- choose the best answer. hydrogen cyanide (hcn) and water (h₂o) are produced from the reaction of methane (ch₄), ammonia (nh₃), and oxygen gas (o₂). which of these best represents the reactants for this reaction? hcn + h₂o + ch₄ ch₄ + nh₃ + o₂ hcn + h₂o ch₄ + nh₃ + o 6) choose all that apply. the following chemical equation shows the formation of lead (ii) chloride. pbo(s) + hcl(aq) → pbcl₂(s) + h₂o(l) which two statements are correct? pbcl₂ and h₂o are the reactants. pbcl₂ and h₂o are the products. pbo and hcl are the products. pbo and hcl are the reactants.
Step1: Identify reactants for Q5
Reactants are the starting substances in a reaction. The problem states HCN and H₂O are products, so the reactants are CH₄, NH₃, and O₂.
Step2: Analyze equation for Q6
In a chemical equation, reactants are on the left of $
ightarrow$, products on the right. For $\text{PbO}(s) + \text{HCl}(aq)
ightarrow \text{PbCl}_2(s) + \text{H}_2\text{O}(l)$:
- Reactants: PbO, HCl
- Products: PbCl₂, H₂O
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 5
$\boldsymbol{\text{CH}_4 + \text{NH}_3 + \text{O}_2}$
Question 6
$\boldsymbol{\text{PbCl}_2 \text{ and } \text{H}_2\text{O are the products.}}$
$\boldsymbol{\text{PbO and HCl are the reactants.}}$