QUESTION IMAGE
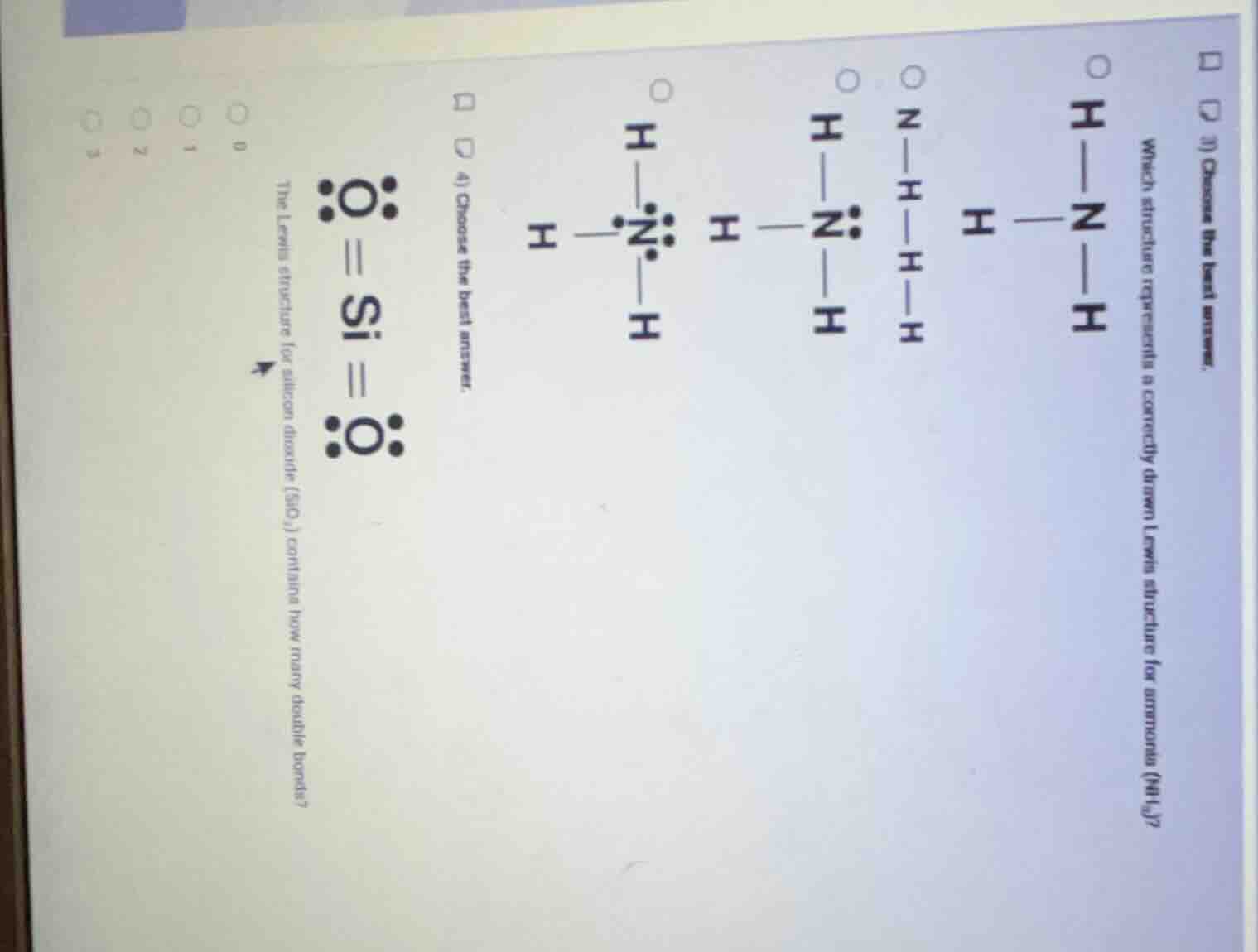
Question
- choose the best answer. which structure represents a correctly drawn lewis structure for ammonia (nh₃)? 4) choose the best answer. the lewis structure for silicon dioxide (sio₂) contains how many double bonds? 0 1 2 3
Step1: Analyze ammonia ($NH_3$) Lewis structure
Nitrogen has 5 valence electrons. It forms 3 single bonds with H atoms (using 3 electrons), leaving 1 lone pair (2 electrons). The correct structure has 3 N-H single bonds and 2 nonbonding electrons on N.
Step2: Evaluate given structures
- $H—N—H$ (no lone pairs): Incorrect, missing N's lone pair.
- $N—H—H—H$: Incorrect bonding arrangement.
- $H—\ddot{N}—H$ with one H: Incorrect, $NH_3$ has 3 H atoms.
- $H—\ddot{N}—H$ with three H atoms ($\ce{H-\underset{\cdot \cdot}{\overset{H}{N}}-H}$): Correct, matches valence electron count and bonding.
Step3: Analyze $SiO_2$ double bonds
In actual $SiO_2$ (network covalent solid), Si forms 4 single bonds to O atoms, no double bonds. The given Lewis structure with double bonds is not representative of the real structure, so the number of double bonds in the actual compound is 0.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\boldsymbol{H—\ddot{N}—H}$ (with three H atoms, $\boldsymbol{\ce{H-\underset{\cdot \cdot}{\overset{H}{N}}-H}}$)
- $\boldsymbol{0}$