QUESTION IMAGE
Question
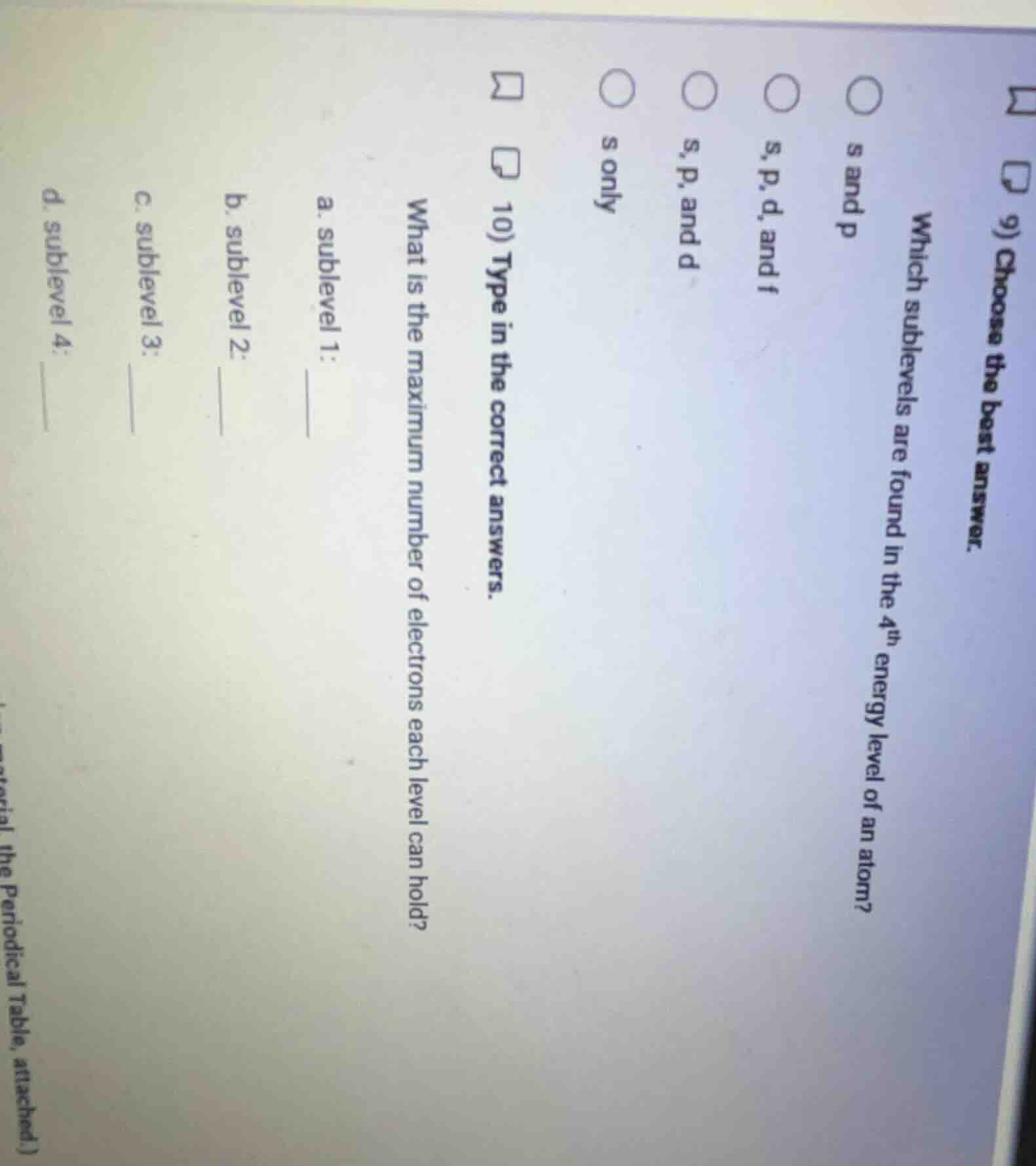
- choose the best answer. which sublevels are found in the 4th energy level of an atom? s and p s, p, d, and f s, p, and d s only 10) type in the correct answers. what is the maximum number of electrons each level can hold? a. sublevel 1: __ b. sublevel 2: c. sublevel 3: d. sublevel 4: __
Step1: Solve question 9
The 4th energy level ($n=4$) can have sublevels corresponding to $l=0,1,2,3$, which are s, p, d, f respectively.
Step2: Solve question 10a
Use electron capacity formula $2n^2$, $n=1$:
$\text{Electrons} = 2(1)^2 = 2$
Step3: Solve question 10b
Use electron capacity formula $2n^2$, $n=2$:
$\text{Electrons} = 2(2)^2 = 8$
Step4: Solve question 10c
Use electron capacity formula $2n^2$, $n=3$:
$\text{Electrons} = 2(3)^2 = 18$
Step5: Solve question 10d
Use electron capacity formula $2n^2$, $n=4$:
$\text{Electrons} = 2(4)^2 = 32$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- s, p, d, and f
10.
a. 2
b. 8
c. 18
d. 32