QUESTION IMAGE
Question
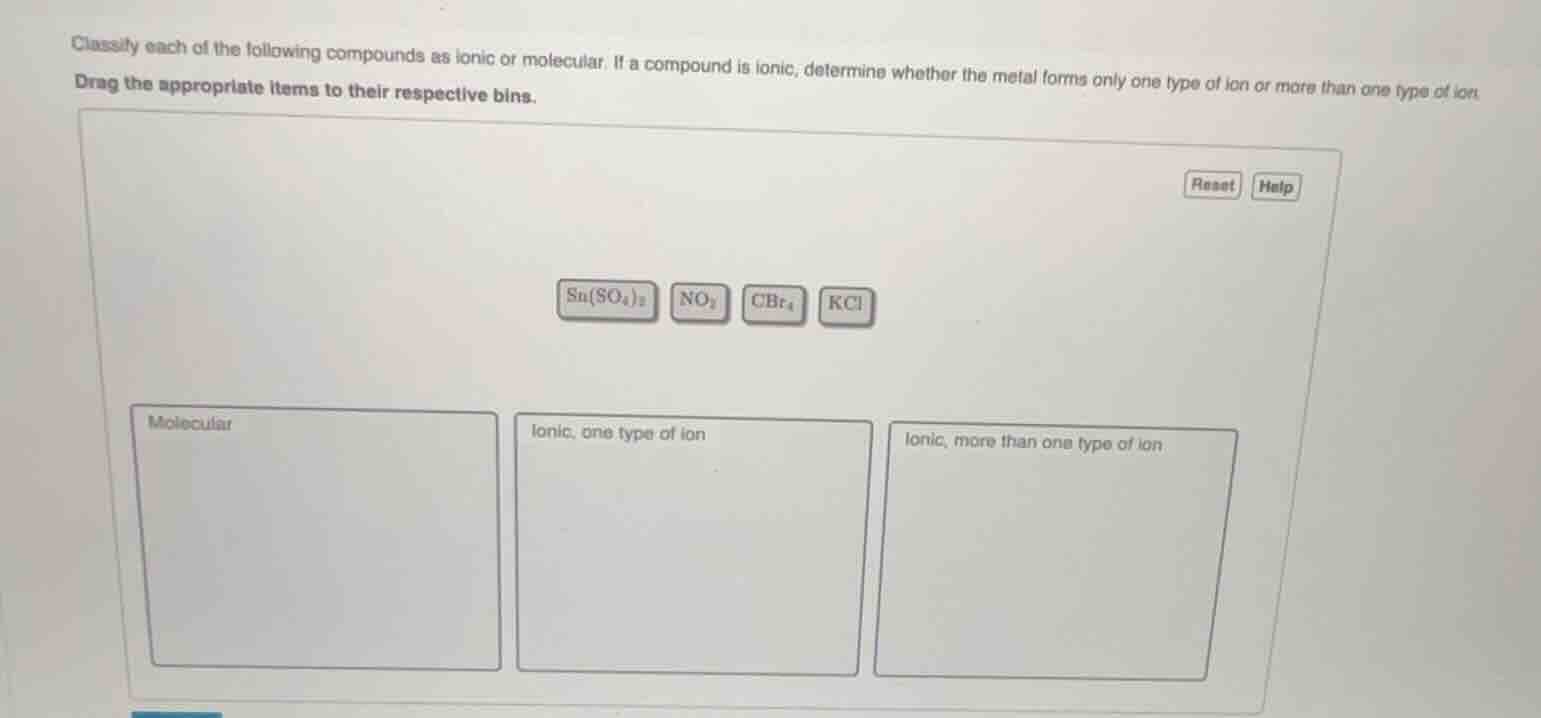
classify each of the following compounds as ionic or molecular. if a compound is ionic, determine whether the metal forms only one type of ion or more than one type of ion. drag the appropriate items to their respective bins. sn(so₄)₂ no₂ cbr₄ kcl molecular ionic, one type of ion ionic, more than one type of ion
Step1: Analyze \( \text{NO}_2 \) and \( \text{CBr}_4 \)
Molecular compounds are formed by non - metals sharing electrons. \( \text{NO}_2 \) (nitrogen and oxygen, both non - metals) and \( \text{CBr}_4 \) (carbon and bromine, both non - metals) are molecular. So they go to the "Molecular" bin.
Step2: Analyze \( \text{KCl} \)
Ionic compounds are formed between metals and non - metals. \( \text{KCl} \) is formed from \( \text{K}^+ \) (potassium ion, potassium is an alkali metal which forms only \( \text{K}^+ \)) and \( \text{Cl}^- \). So \( \text{KCl} \) is ionic with the metal forming one type of ion, and it goes to the "Ionic, one type of ion" bin.
Step3: Analyze \( \text{Sn(SO}_4\text{)}_2 \)
\( \text{Sn(SO}_4\text{)}_2 \) is ionic (formed from \( \text{Sn}^{4 + } \) and \( \text{SO}_4^{2 - } \)). Tin (Sn) can form different ions (e.g., \( \text{Sn}^{2+} \) and \( \text{Sn}^{4+} \)), so the metal forms more than one type of ion. Thus, \( \text{Sn(SO}_4\text{)}_2 \) goes to the "Ionic, more than one type of ion" bin.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Molecular: \( \text{NO}_2 \), \( \text{CBr}_4 \)
- Ionic, one type of ion: \( \text{KCl} \)
- Ionic, more than one type of ion: \( \text{Sn(SO}_4\text{)}_2 \)