QUESTION IMAGE
Question
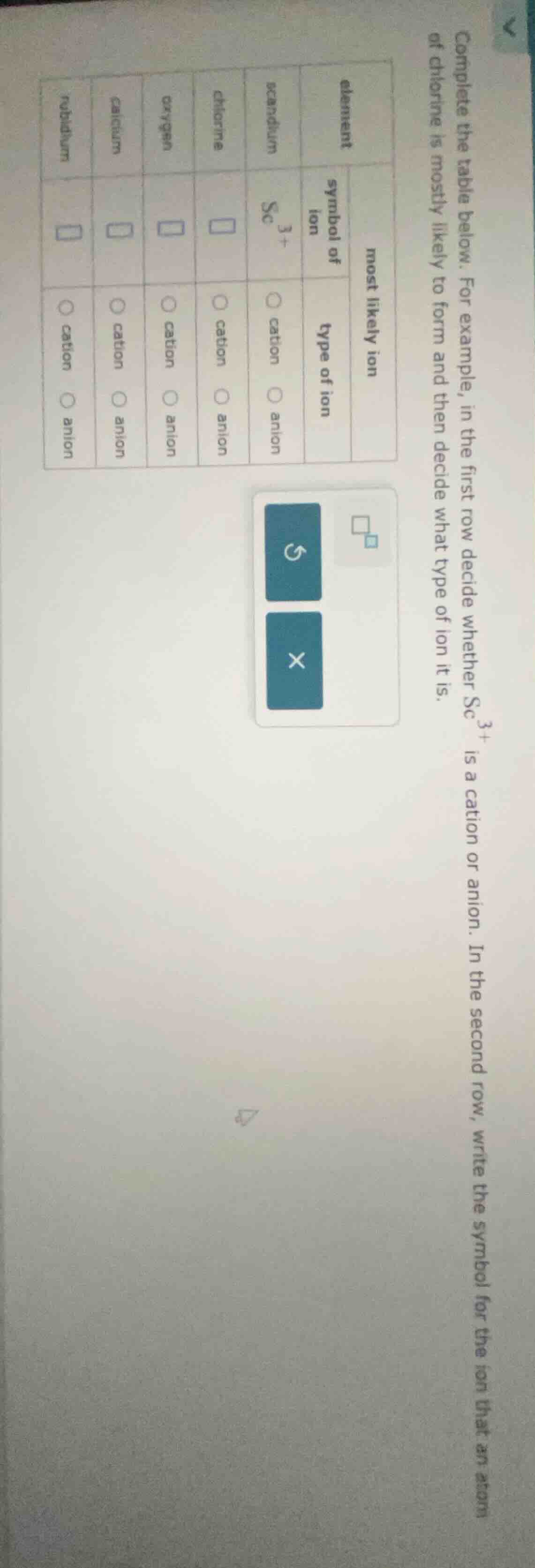
complete the table below. for example, in the first row decide whether $ce{sc^{3+}}$ is a cation or anion. in the second row, write the symbol for the ion that an atom of chlorine is mostly likely to form and then decide what type of ion it is.
| most likely ion | element | symbol of ion | type of ion |
|---|---|---|---|
| chlorine | $square$ | $circ$ cation $circ$ anion | |
| oxygen | $square$ | $circ$ cation $circ$ anion | |
| calcium | $square$ | $circ$ cation $circ$ anion | |
| rubidium | $square$ | $circ$ cation $circ$ anion |
Brief Explanations
- Scandium (metal) loses 3 electrons → $\text{Sc}^{3+}$ (cation).
- Chlorine (nonmetal) gains 1 electron → $\text{Cl}^{-}$ (anion).
- Oxygen (nonmetal) gains 2 electrons → $\text{O}^{2-}$ (anion).
- Calcium (metal) loses 2 electrons → $\text{Ca}^{2+}$ (cation).
- Rubidium (metal) loses 1 electron → $\text{Rb}^{+}$ (cation).
Cations are positive ions (lose electrons); anions are negative ions (gain electrons). Metals form cations, nonmetals form anions.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Element | Symbol of most likely ion | Type of ion |
|---|---|---|
| Chlorine | $\text{Cl}^{-}$ | anion |
| Oxygen | $\text{O}^{2-}$ | anion |
| Calcium | $\text{Ca}^{2+}$ | cation |
| Rubidium | $\text{Rb}^{+}$ | cation |