QUESTION IMAGE
Question
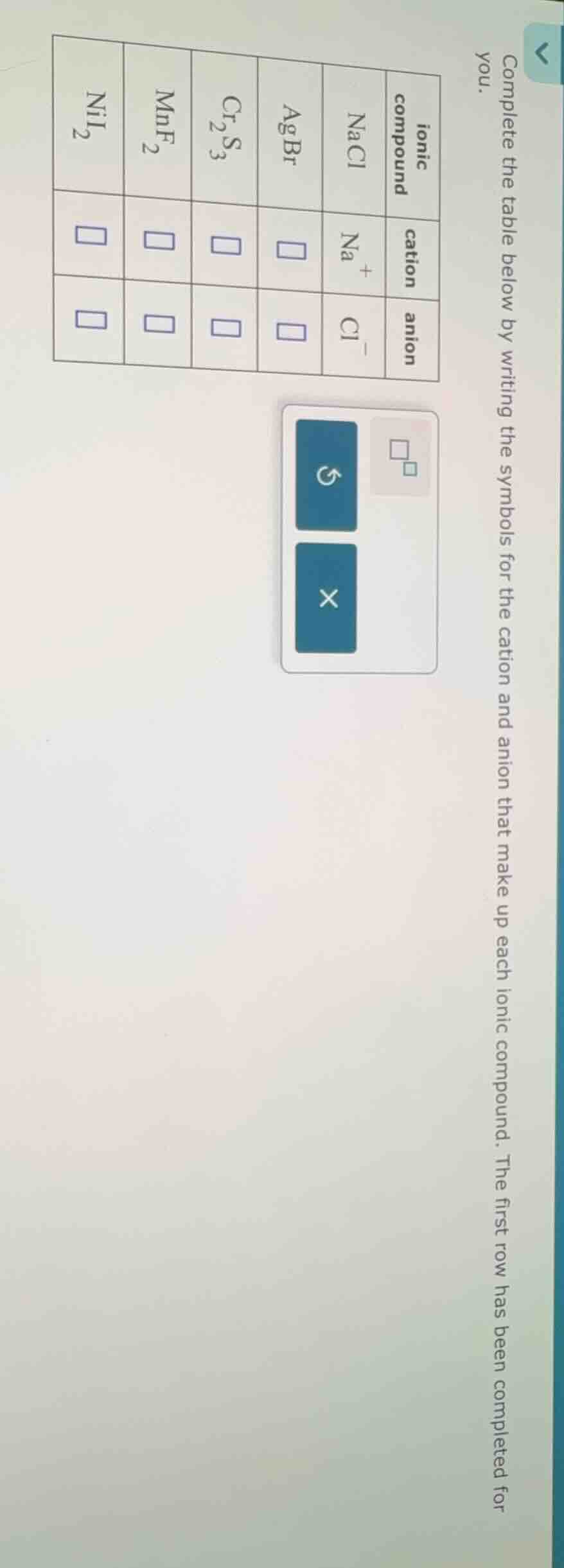
complete the table below by writing the symbols for the cation and anion that make up each ionic compound. the first row has been completed for you.
| ionic compound | cation | anion |
|---|---|---|
| agbr | $square$ | $square$ |
| $ce{cr2s3}$ | $square$ | $square$ |
| $ce{mnf2}$ | $square$ | $square$ |
| $ce{nii2}$ | $square$ | $square$ |
Step1: Analyze AgBr
Ionic compound AgBr is formed by silver ion and bromide ion. Silver has a +1 charge, bromide has -1. So cation is $\text{Ag}^+$, anion is $\text{Br}^-$.
Step2: Analyze $\text{Cr}_2\text{S}_3$
In $\text{Cr}_2\text{S}_3$, let Cr charge be $x$. $2x + 3(-2)=0\Rightarrow x = +3$. So cation is $\text{Cr}^{3+}$, anion is $\text{S}^{2-}$ (sulfide ion, charge -2).
Step3: Analyze $\text{MnF}_2$
For $\text{MnF}_2$, let Mn charge be $y$. $y + 2(-1)=0\Rightarrow y = +2$. Cation is $\text{Mn}^{2+}$, anion is $\text{F}^-$ (fluoride ion, charge -1).
Step4: Analyze $\text{NiI}_2$
In $\text{NiI}_2$, let Ni charge be $z$. $z + 2(-1)=0\Rightarrow z = +2$. Cation is $\text{Ni}^{2+}$, anion is $\text{I}^-$ (iodide ion, charge -1).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Ionic Compound | Cation | Anion |
|---|---|---|
| $\text{Cr}_2\text{S}_3$ | $\text{Cr}^{3+}$ | $\text{S}^{2-}$ |
| $\text{MnF}_2$ | $\text{Mn}^{2+}$ | $\text{F}^-$ |
| $\text{NiI}_2$ | $\text{Ni}^{2+}$ | $\text{I}^-$ |