QUESTION IMAGE
Question
in the compound represented by the chemical formula nh₄cl there would be...
select one:
a. 1 carbon atom.
b. 4 chlorine atoms.
c. 1 hydrogen atom.
d. 1 nitrogen atom.
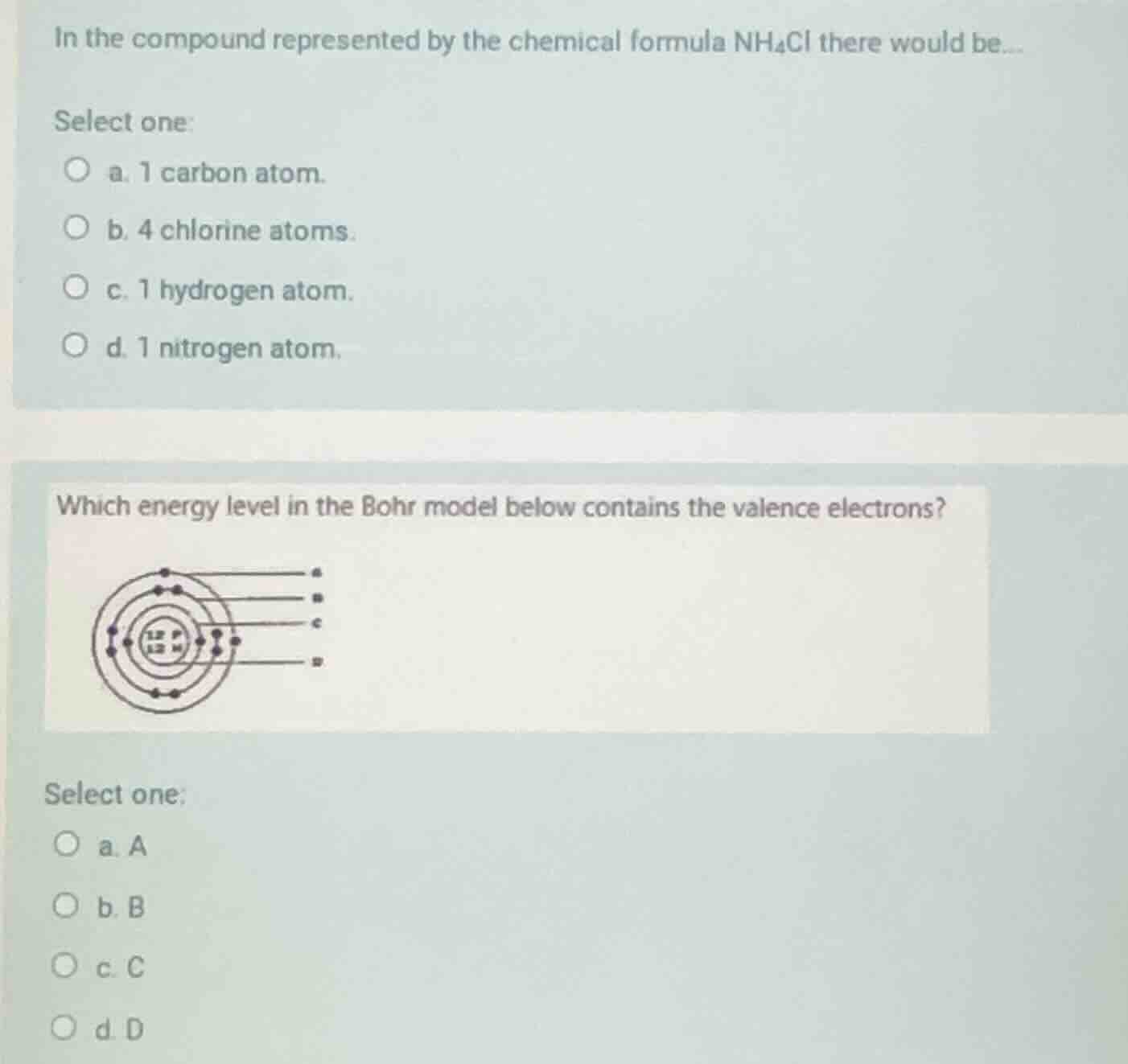
which energy level in the bohr model below contains the valence electrons?
image omitted
select one:
a. a
b. b
c. c
d. d
Step1: Analyze $\text{NH}_4\text{Cl}$ atom counts
Break down the chemical formula: $\text{N}$ (nitrogen) has no subscript = 1 atom; $\text{H}$ has subscript 4 = 4 atoms; $\text{Cl}$ (chlorine) has no subscript = 1 atom; no $\text{C}$ (carbon) present.
Step2: Identify valence energy level
Valence electrons occupy the outermost (highest) energy level in a Bohr model, which is the outermost ring labeled A.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- d. 1 nitrogen atom.
- a. A