QUESTION IMAGE
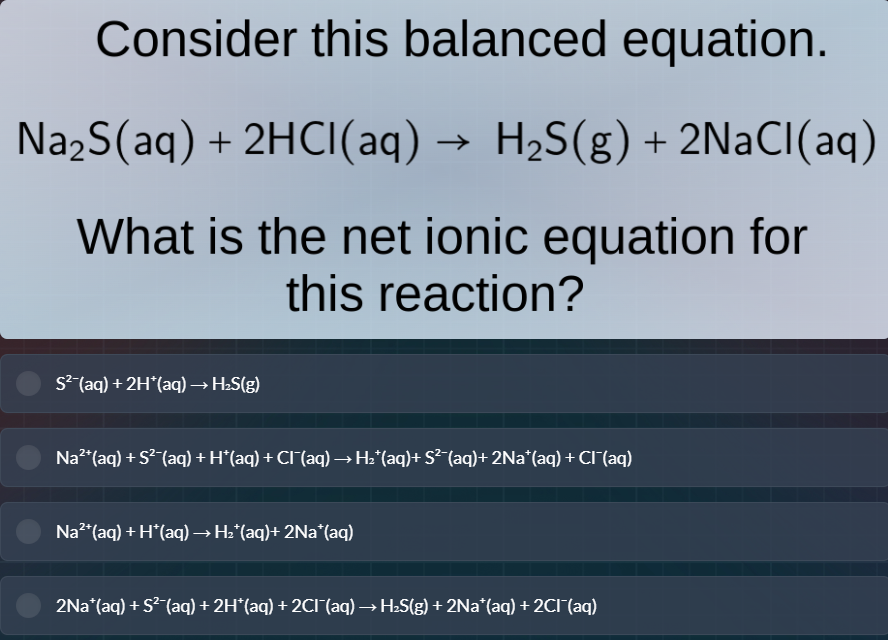
Question
consider this balanced equation.$ce{na_{2}s(aq) + 2hcl(aq) -> h_{2}s(g) + 2nacl(aq)}$what is the net ionic equation for this reaction?$ce{s^{2-}(aq) + 2h^{+}(aq) -> h_{2}s(g)}$$ce{na^{2+}(aq) + s^{2-}(aq) + h^{+}(aq) + cl^{-}(aq) -> h_{2}^{+}(aq) + s^{2-}(aq) + 2na^{+}(aq) + cl^{-}(aq)}$$ce{na^{2+}(aq) + h^{+}(aq) -> h_{2}^{+}(aq) + 2na^{+}(aq)}$$ce{2na^{+}(aq) + s^{2-}(aq) + 2h^{+}(aq) + 2cl^{-}(aq) -> h_{2}s(g) + 2na^{+}(aq) + 2cl^{-}(aq)}$
Step1: Split into ionic components
$\text{Na}_2\text{S}(aq)
ightarrow 2\text{Na}^+(aq) + \text{S}^{2-}(aq)$
$2\text{HCl}(aq)
ightarrow 2\text{H}^+(aq) + 2\text{Cl}^-(aq)$
$2\text{NaCl}(aq)
ightarrow 2\text{Na}^+(aq) + 2\text{Cl}^-(aq)$
$\text{H}_2\text{S}(g)$ remains as is (gas, not dissociated)
Full ionic equation:
$2\text{Na}^+(aq) + \text{S}^{2-}(aq) + 2\text{H}^+(aq) + 2\text{Cl}^-(aq)
ightarrow \text{H}_2\text{S}(g) + 2\text{Na}^+(aq) + 2\text{Cl}^-(aq)$
Step2: Cancel spectator ions
Spectator ions: $\text{Na}^+(aq)$, $\text{Cl}^-(aq)$ (present on both sides)
Cancel them from both sides:
$\text{S}^{2-}(aq) + 2\text{H}^+(aq)
ightarrow \text{H}_2\text{S}(g)$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{S}^{2-}(aq) + 2\text{H}^+(aq)
ightarrow \text{H}_2\text{S}(g)$ (Option 1)