QUESTION IMAGE
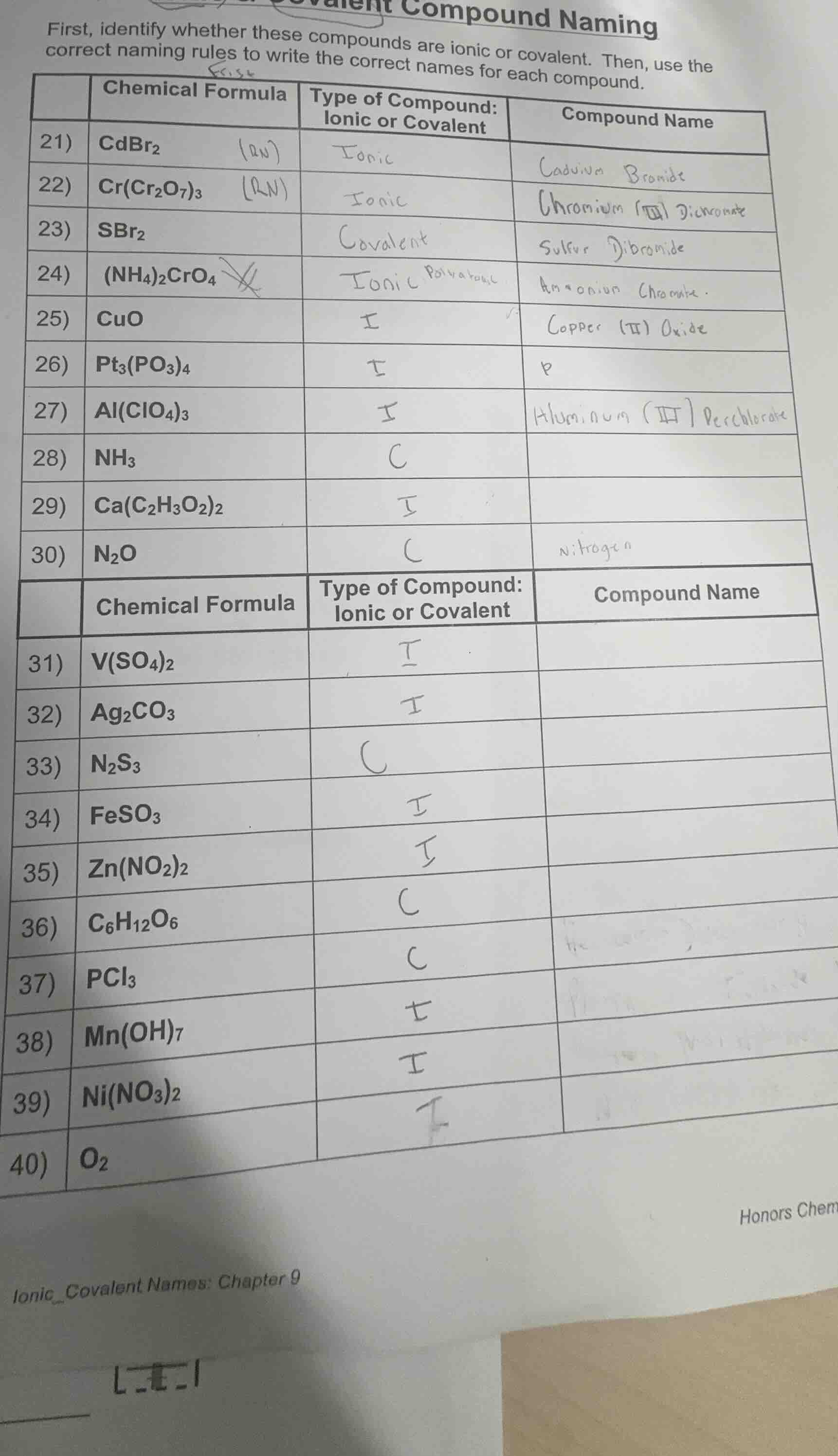
Question
covalent compound naming
first, identify whether these compounds are ionic or covalent. then, use the
correct naming rules to write the correct names for each compound.
| chemical formula | type of compound: |
ionic or covalent | compound name
- | cdbr₂ | ionic | cadmium bromide
- | cr(cr₂o₇)₃ | ionic | chromium (iii) dichromate
- | sbr₂ | covalent | sulfur dibromide
- | (nh₄)₂cro₄ | ionic polyatomic | ammonium chromate
- | cuo | i | copper (ii) oxide
- | pt₃(po₃)₄ | i |
- | al(clo₄)₃ | i | aluminum (iii) perchlorate
- | nh₃ | c |
- | ca(c₂h₃o₂)₂ | i |
- | n₂o | c | nitrogen
| chemical formula | type of compound: |
ionic or covalent | compound name
- | v(so₄)₂ | i |
- | ag₂co₃ | i |
- | n₂s₃ | c |
- | feso₃ | i |
- | zn(no₂)₂ | i |
- | c₆h₁₂o₆ | c |
- | pcl₃ | c |
- | mn(oh)₇ | i |
- | ni(no₃)₂ | i |
- | o₂ | |
ionic_covalent names: chapter 9
honors chem
Step1: Verify compound type & name
Check existing entries, correct errors, complete blanks.
Step2: Ionic: Name cation + anion (charge if needed)
For ionic compounds, identify cation/anion, balance charges.
Step3: Covalent: Prefix + element + prefix + ide
For covalent, use Greek prefixes for atom counts.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| # | Chemical Formula | Type of Compound: Ionic or Covalent | Compound Name |
|---|---|---|---|
| 22) | $\text{Cr(Cr}_2\text{O}_7\text{)}_3$ | Ionic | Chromium(III) Dichromate |
| 23) | $\text{SBr}_2$ | Covalent | Sulfur Dibromide |
| 24) | $\text{(NH}_4\text{)}_2\text{CrO}_4$ | Ionic | Ammonium Chromate |
| 25) | $\text{CuO}$ | Ionic | Copper(II) Oxide |
| 26) | $\text{Pt}_3\text{(PO}_3\text{)}_4$ | Ionic | Platinum(IV) Phosphite |
| 27) | $\text{Al(ClO}_4\text{)}_3$ | Ionic | Aluminum Perchlorate |
| 28) | $\text{NH}_3$ | Covalent | Ammonia (or Nitrogen Trihydride) |
| 29) | $\text{Ca(C}_2\text{H}_3\text{O}_2\text{)}_2$ | Ionic | Calcium Acetate |
| 30) | $\text{N}_2\text{O}$ | Covalent | Dinitrogen Monoxide |
| 31) | $\text{V(SO}_4\text{)}_2$ | Ionic | Vanadium(IV) Sulfate |
| 32) | $\text{Ag}_2\text{CO}_3$ | Ionic | Silver Carbonate |
| 33) | $\text{N}_2\text{S}_3$ | Covalent | Dinitrogen Trisulfide |
| 34) | $\text{FeSO}_3$ | Ionic | Iron(II) Sulfite |
| 35) | $\text{Zn(NO}_2\text{)}_2$ | Ionic | Zinc Nitrite |
| 36) | $\text{C}_6\text{H}_{12}\text{O}_6$ | Covalent | Glucose (or Hexose, or Dextrose) |
| 37) | $\text{PCl}_3$ | Covalent | Phosphorus Trichloride |
| 38) | $\text{Mn(OH)}_7$ | Ionic | Manganese(VII) Hydroxide |
| 39) | $\text{Ni(NO}_3\text{)}_2$ | Ionic | Nickel(II) Nitrate |
| 40) | $\text{O}_2$ | Covalent | Diatomic Oxygen (or Oxygen Gas) |