QUESTION IMAGE
Question
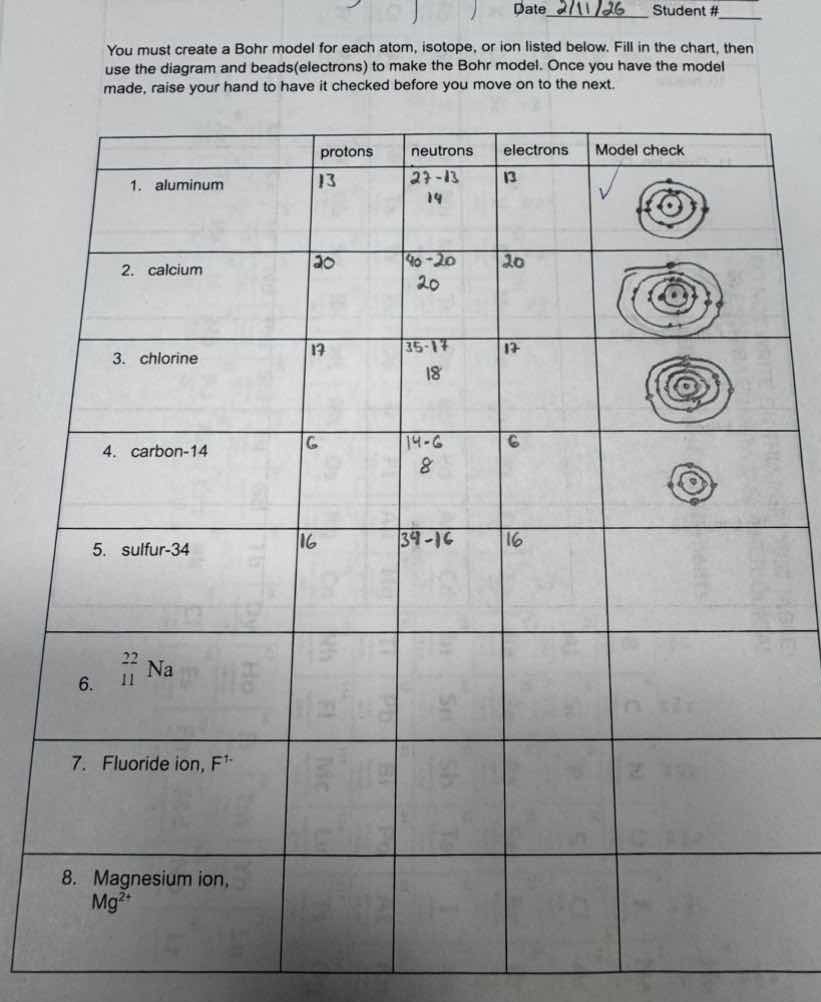
date 2/11/26 student #____you must create a bohr model for each atom, isotope, or ion listed below. fill in the chart, then use the diagram and beads(electrons) to make the bohr model. once you have the model made, raise your hand to have it checked before you move on to the next.| | protons | neutrons | electrons | model check || ---- | ---- | ---- | ---- | ---- || 1. aluminum | 13 | 27-13<br>14 | 13 | ✓ || 2. calcium | 20 | 40-20<br>20 | 20 | || 3. chlorine | 17 | 35-17<br>18 | 17 | || 4. carbon-14 | 6 | 14-6<br>8 | 6 | || 5. sulfur-34 | 16 | 34-16 | 16 | || 6. $^{22}_{11}$na | | | | || 7. fluoride ion, f⁻ | | | | || 8. magnesium ion, mg²⁺ | | | | |
Step1: Recall atomic number rules
For any atom/ion:
- Protons = Atomic number ($Z$)
- Neutrons = Mass number ($A$) - Atomic number ($Z$)
- Neutral atoms: Electrons = Protons
- Negative ions: Electrons = Protons + charge magnitude
- Positive ions: Electrons = Protons - charge magnitude
---
Step2: Solve for $^{22}_{11}\text{Na}$
Protons = Atomic number = $11$
Neutrons = $22 - 11 = 11$
Neutral atom, so Electrons = $11$
<Expression>
$\text{Protons}=11, \text{Neutrons}=22-11=11, \text{Electrons}=11$
</Expression>
Step3: Solve for Fluoride ion ($\text{F}^-$)
Fluorine atomic number = $9$
Protons = $9$
Assume most common isotope ($A=19$): Neutrons = $19 - 9 = 10$
Negative ion ($-1$ charge): Electrons = $9 + 1 = 10$
<Expression>
$\text{Protons}=9, \text{Neutrons}=19-9=10, \text{Electrons}=9+1=10$
</Expression>
Step4: Solve for Magnesium ion ($\text{Mg}^{2+}$)
Magnesium atomic number = $12$
Protons = $12$
Assume most common isotope ($A=24$): Neutrons = $24 - 12 = 12$
Positive ion ($+2$ charge): Electrons = $12 - 2 = 10$
<Expression>
$\text{Protons}=12, \text{Neutrons}=24-12=12, \text{Electrons}=12-2=10$
</Expression>
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| protons | neutrons | electrons | |
|---|---|---|---|
| 7. Fluoride ion, $\text{F}^-$ | 9 | 10 | 10 |
| 8. Magnesium ion, $\text{Mg}^{2+}$ | 12 | 12 | 10 |
*(Note: For the Bohr models:
- $^{22}_{11}\text{Na}$: 2 electrons in first shell, 8 in second, 1 in third
- $\text{F}^-$: 2 electrons in first shell, 8 in second
- $\text{Mg}^{2+}$: 2 electrons in first shell, 8 in second)*