QUESTION IMAGE
Question
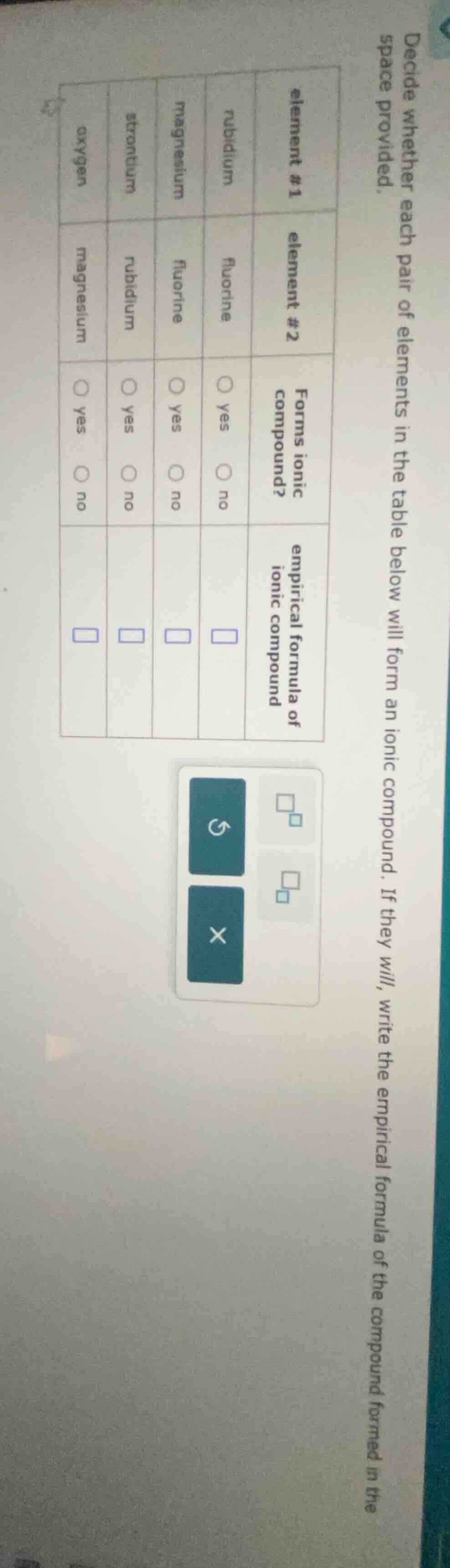
decide whether each pair of elements in the table below will form an ionic compound. if they will, write the empirical formula of the compound formed in the space provided.
| element #1 | element #2 | forms ionic compound? | empirical formula of ionic compound |
|---|---|---|---|
| magnesium | fluorine | ∘ yes ∘ no | $square$ |
| strontium | rubidium | ∘ yes ∘ no | $square$ |
| oxygen | magnesium | ∘ yes ∘ no | $square$ |
Response
To solve this, we analyze each pair:
Pair 1: Rubidium (Rb) & Fluorine (F)
- Rubidium is a metal (alkali metal, Group 1), Fluorine is a non - metal (halogen, Group 17). Metals and non - metals form ionic compounds.
- Rubidium has a +1 charge (\(Rb^+\)), Fluorine has a -1 charge (\(F^-\)). The formula is \(RbF\) (since \(1(+1)+1(-1)=0\)).
Pair 2: Magnesium (Mg) & Fluorine (F)
- Magnesium is a metal (alkaline earth metal, Group 2), Fluorine is a non - metal. They form an ionic compound.
- Magnesium has a +2 charge (\(Mg^{2+}\)), Fluorine has a -1 charge (\(F^-\)). To balance charges, we need 2 \(F^-\) for 1 \(Mg^{2+}\) (since \(2(+1)+1(+2)= + 4\) is wrong, correct: \(1(+2)+2(-1)=0\)). The formula is \(MgF_2\).
Pair 3: Strontium (Sr) & Rubidium (Rb)
- Both are metals. Metals do not form ionic compounds with other metals (ionic compounds require a metal and a non - metal). So, they do not form an ionic compound.
Pair 4: Oxygen (O) & Magnesium (Mg)
- Magnesium is a metal, Oxygen is a non - metal. They form an ionic compound.
- Magnesium has a +2 charge (\(Mg^{2+}\)), Oxygen has a -2 charge (\(O^{2-}\)). The formula is \(MgO\) (since \(1(+2)+1(-2)=0\)).
Final Answers (for “Forms ionic compound?” and formula):
| Element #1 | Element #2 | Forms ionic compound? | Empirical formula of ionic compound |
|---|---|---|---|
| Magnesium | Fluorine | Yes | \(MgF_2\) |
| Strontium | Rubidium | No | - |
| Oxygen | Magnesium | Yes | \(MgO\) |
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this, we analyze each pair:
Pair 1: Rubidium (Rb) & Fluorine (F)
- Rubidium is a metal (alkali metal, Group 1), Fluorine is a non - metal (halogen, Group 17). Metals and non - metals form ionic compounds.
- Rubidium has a +1 charge (\(Rb^+\)), Fluorine has a -1 charge (\(F^-\)). The formula is \(RbF\) (since \(1(+1)+1(-1)=0\)).
Pair 2: Magnesium (Mg) & Fluorine (F)
- Magnesium is a metal (alkaline earth metal, Group 2), Fluorine is a non - metal. They form an ionic compound.
- Magnesium has a +2 charge (\(Mg^{2+}\)), Fluorine has a -1 charge (\(F^-\)). To balance charges, we need 2 \(F^-\) for 1 \(Mg^{2+}\) (since \(2(+1)+1(+2)= + 4\) is wrong, correct: \(1(+2)+2(-1)=0\)). The formula is \(MgF_2\).
Pair 3: Strontium (Sr) & Rubidium (Rb)
- Both are metals. Metals do not form ionic compounds with other metals (ionic compounds require a metal and a non - metal). So, they do not form an ionic compound.
Pair 4: Oxygen (O) & Magnesium (Mg)
- Magnesium is a metal, Oxygen is a non - metal. They form an ionic compound.
- Magnesium has a +2 charge (\(Mg^{2+}\)), Oxygen has a -2 charge (\(O^{2-}\)). The formula is \(MgO\) (since \(1(+2)+1(-2)=0\)).
Final Answers (for “Forms ionic compound?” and formula):
| Element #1 | Element #2 | Forms ionic compound? | Empirical formula of ionic compound |
|---|---|---|---|
| Magnesium | Fluorine | Yes | \(MgF_2\) |
| Strontium | Rubidium | No | - |
| Oxygen | Magnesium | Yes | \(MgO\) |