QUESTION IMAGE
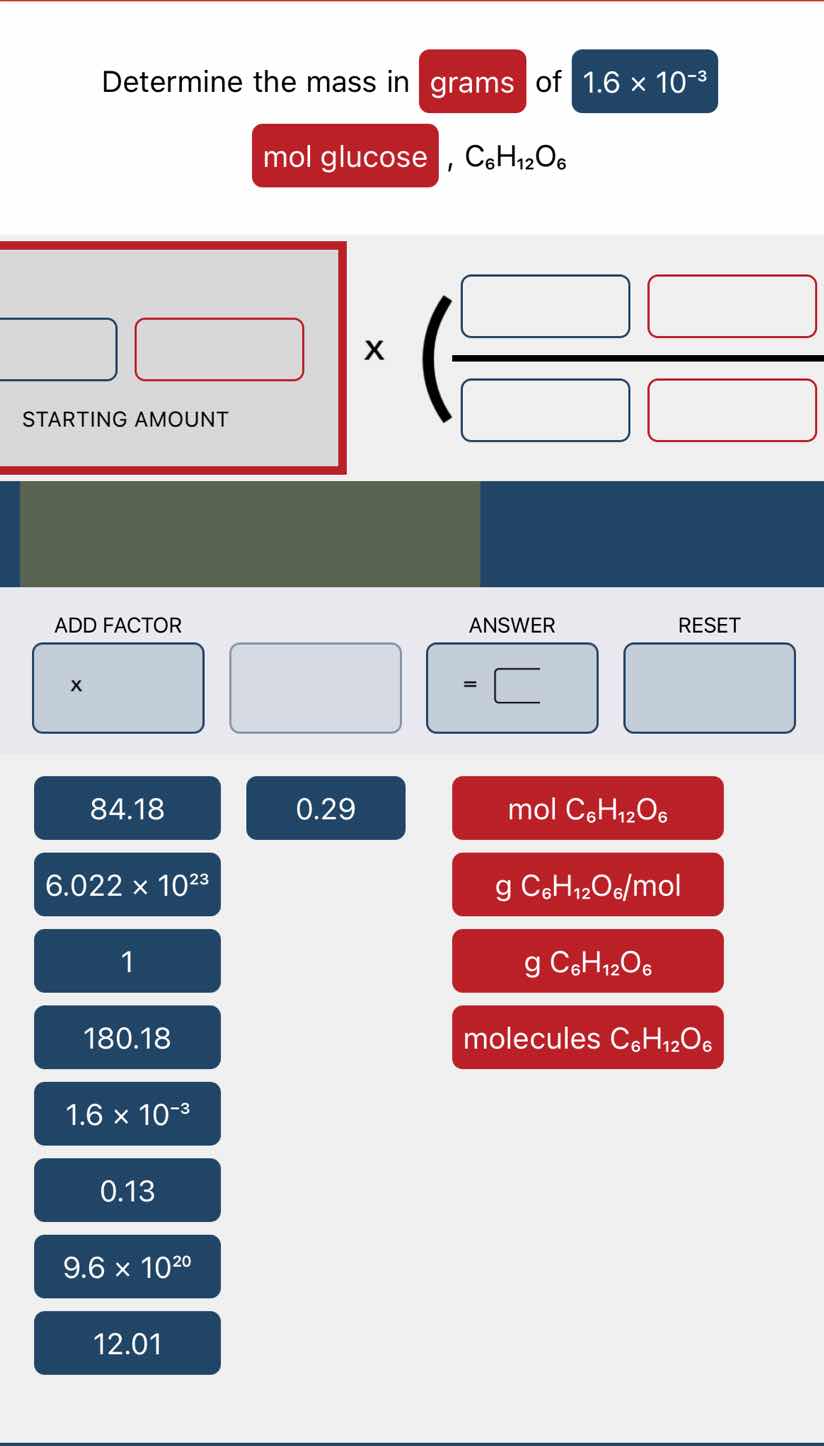
Question
determine the mass in grams of 1.6 × 10⁻³ mol glucose, c₆h₁₂o₆. the interface has a starting amount section, a calculation formula section, and buttons: add factor (with x button), answer (with = and a box), reset. below are factor buttons: blue (84.18, 0.29, 6.022 × 10²³, 1, 180.18, 1.6 × 10⁻³, 0.13, 9.6 × 10²⁰, 12.01) and red (mol c₆h₁₂o₆, g c₆h₁₂o₆/mol, g c₆h₁₂o₆, molecules c₆h₁₂o₆).
Step1: Identify starting amount
Starting amount: $1.6 \times 10^{-3}\ \text{mol } C_6H_{12}O_6$
Step2: Calculate molar mass of glucose
Molar mass of $C_6H_{12}O_6$:
$$(6 \times 12.01) + (12 \times 1.008) + (6 \times 16.00) = 180.18\ \text{g/mol}$$
Step3: Convert moles to grams
Multiply moles by molar mass:
$$1.6 \times 10^{-3}\ \text{mol} \times 180.18\ \frac{\text{g}}{\text{mol}}$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$0.29\ \text{g}$
(For the interactive template:
- Starting amount box: $1.6 \times 10^{-3}$ and $\text{mol } C_6H_{12}O_6$
- Conversion factor numerator: $180.18$ and $\text{g } C_6H_{12}O_6$
- Conversion factor denominator: $1$ and $\text{mol } C_6H_{12}O_6$)