QUESTION IMAGE
Question
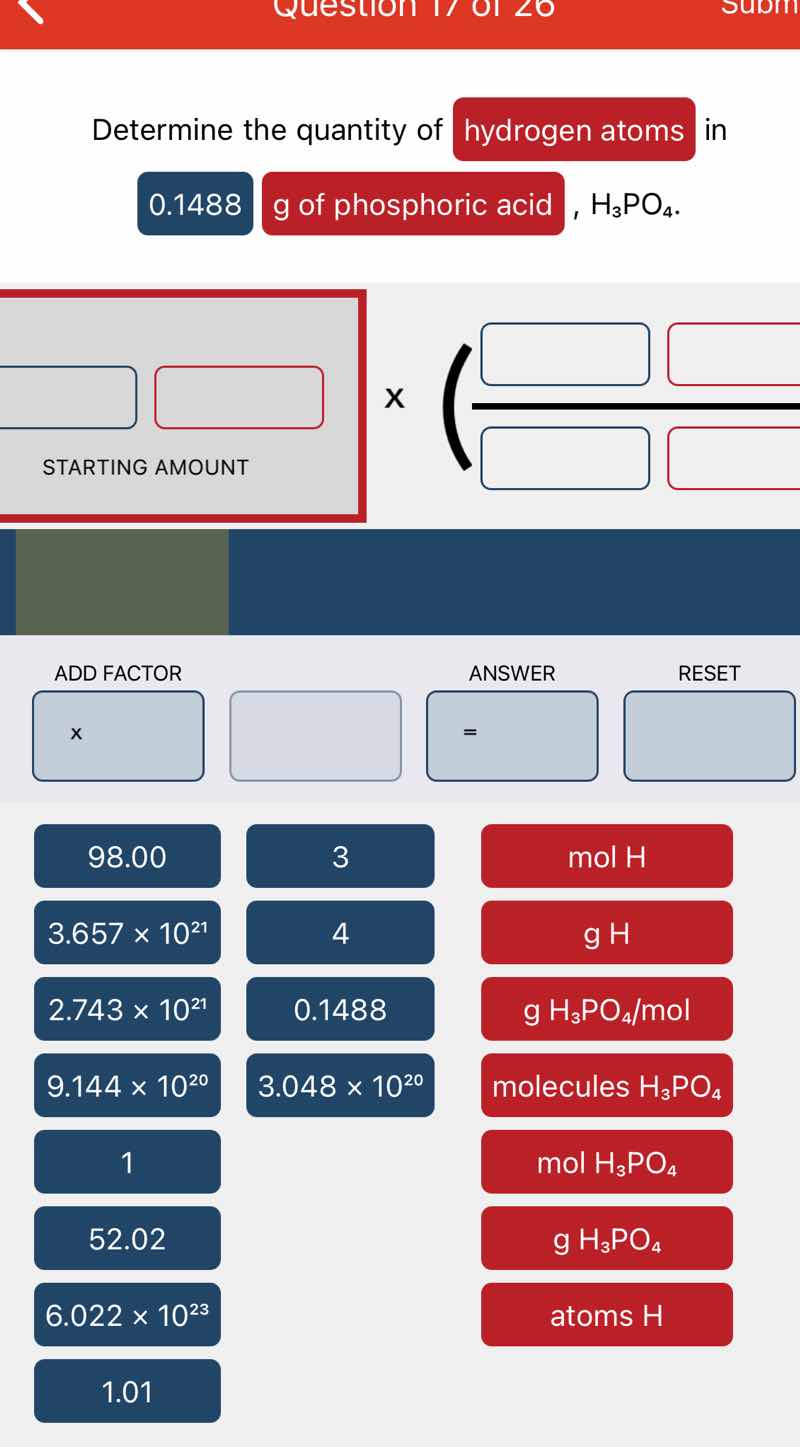
determine the quantity of hydrogen atoms in 0.1488 g of phosphoric acid, h₃po₄.
(starting amount section, then a multiplication with factors, and a set of buttons with values like 98.00, 3, 3.657×10²¹, 4, 2.743×10²¹, 0.1488, 9.144×10²⁰, 3.048×10²⁰, 1, 52.02, 6.022×10²³, 1.01 and units like mol h, g h, g h₃po₄/mol, molecules h₃po₄, mol h₃po₄, g h₃po₄, atoms h)
Step1: Start with given mass
$0.1488\ \text{g}\ \text{H}_3\text{PO}_4$
Step2: Convert to moles of $\text{H}_3\text{PO}_4$
Multiply by $\frac{1\ \text{mol}\ \text{H}_3\text{PO}_4}{98.00\ \text{g}\ \text{H}_3\text{PO}_4}$
Expression: $0.1488\ \text{g}\ \text{H}_3\text{PO}_4 \times \frac{1\ \text{mol}\ \text{H}_3\text{PO}_4}{98.00\ \text{g}\ \text{H}_3\text{PO}_4}$
Step3: Convert to moles of H
Multiply by $\frac{3\ \text{mol}\ \text{H}}{1\ \text{mol}\ \text{H}_3\text{PO}_4}$
Expression: $0.1488 \times \frac{1}{98.00} \times \frac{3\ \text{mol}\ \text{H}}{1\ \text{mol}\ \text{H}_3\text{PO}_4}$
Step4: Convert to atoms of H
Multiply by $\frac{6.022 \times 10^{23}\ \text{atoms H}}{1\ \text{mol H}}$
Expression: $0.1488 \times \frac{1}{98.00} \times 3 \times 6.022 \times 10^{23}$
Step5: Calculate final value
$\frac{0.1488 \times 3 \times 6.022 \times 10^{23}}{98.00} = 2.743 \times 10^{21}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$2.743 \times 10^{21}$ atoms H