QUESTION IMAGE
Question
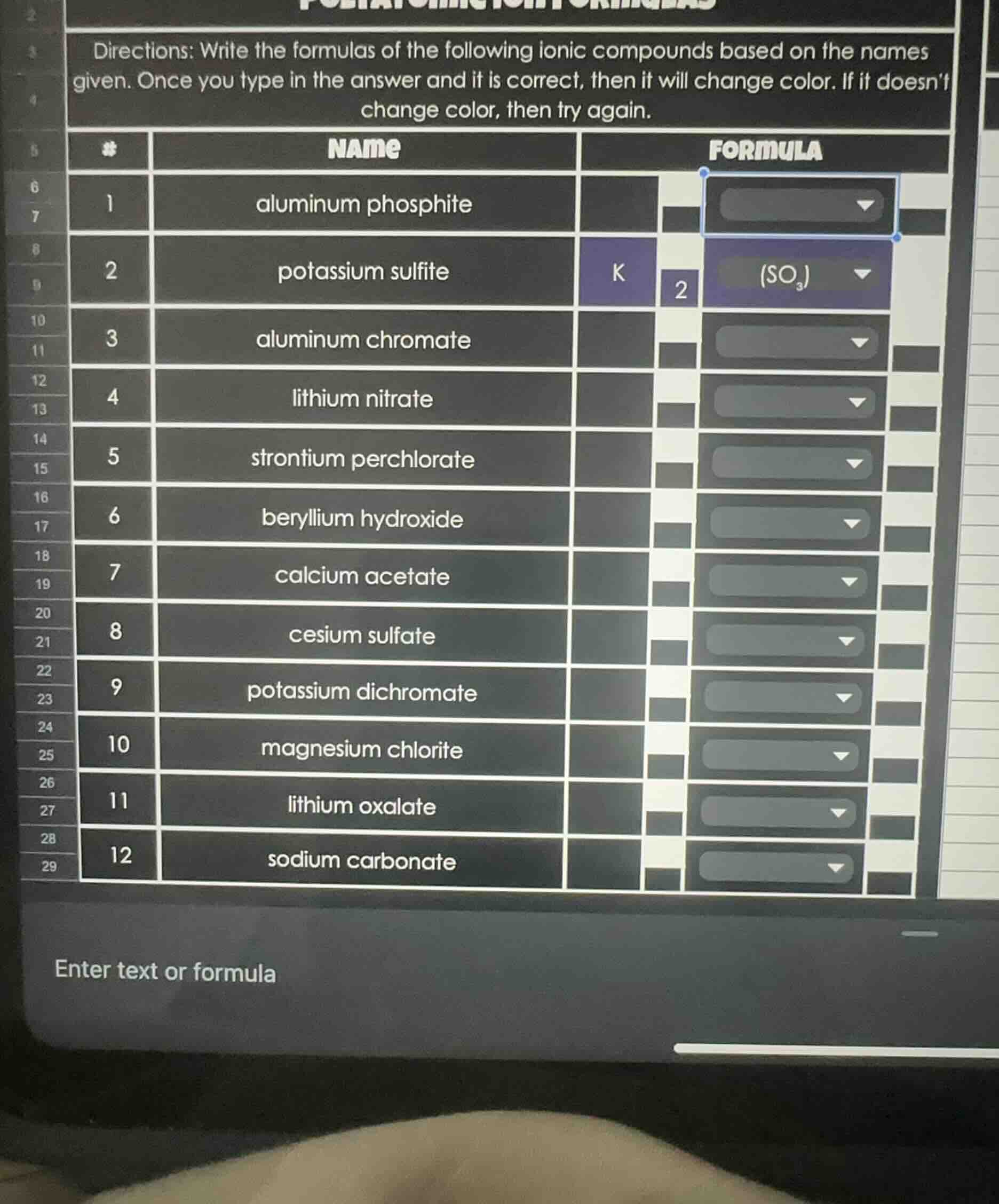
directions: write the formulas of the following ionic compounds based on the names given. once you type in the answer and it is correct, then it will change color. if it doesn’t change color, then try again.
| name | formula
1 | aluminum phosphite |
2 | potassium sulfite | k₂(so₃)
3 | aluminum chromate |
4 | lithium nitrate |
5 | strontium perchlorate |
6 | beryllium hydroxide |
7 | calcium acetate |
8 | cesium sulfate |
9 | potassium dichromate |
10 | magnesium chlorite |
11 | lithium oxalate |
12 | sodium carbonate |
Step1: Identify ion charges (aluminum phosphite)
Aluminum: $\text{Al}^{3+}$, Phosphite: $\text{PO}_3^{3-}$
Step2: Balance charges (aluminum phosphite)
$\text{Al}^{3+} + \text{PO}_3^{3-}
ightarrow \text{AlPO}_3$
Step3: Correct potassium sulfite formula
Potassium: $\text{K}^+$, Sulfite: $\text{SO}_3^{2-}$; balance: $2\text{K}^+ + \text{SO}_3^{2-}
ightarrow \text{K}_2\text{SO}_3$
Step4: Identify ion charges (aluminum chromate)
Aluminum: $\text{Al}^{3+}$, Chromate: $\text{CrO}_4^{2-}$
Step5: Balance charges (aluminum chromate)
$2\text{Al}^{3+} + 3\text{CrO}_4^{2-}
ightarrow \text{Al}_2(\text{CrO}_4)_3$
Step6: Identify ion charges (lithium nitrate)
Lithium: $\text{Li}^+$, Nitrate: $\text{NO}_3^-$
Step7: Balance charges (lithium nitrate)
$\text{Li}^+ + \text{NO}_3^-
ightarrow \text{LiNO}_3$
Step8: Identify ion charges (strontium perchlorate)
Strontium: $\text{Sr}^{2+}$, Perchlorate: $\text{ClO}_4^-$
Step9: Balance charges (strontium perchlorate)
$\text{Sr}^{2+} + 2\text{ClO}_4^-
ightarrow \text{Sr}(\text{ClO}_4)_2$
Step10: Identify ion charges (beryllium hydroxide)
Beryllium: $\text{Be}^{2+}$, Hydroxide: $\text{OH}^-$
Step11: Balance charges (beryllium hydroxide)
$\text{Be}^{2+} + 2\text{OH}^-
ightarrow \text{Be}(\text{OH})_2$
Step12: Identify ion charges (calcium acetate)
Calcium: $\text{Ca}^{2+}$, Acetate: $\text{C}_2\text{H}_3\text{O}_2^-$
Step13: Balance charges (calcium acetate)
$\text{Ca}^{2+} + 2\text{C}_2\text{H}_3\text{O}_2^-
ightarrow \text{Ca}(\text{C}_2\text{H}_3\text{O}_2)_2$
Step14: Identify ion charges (cesium sulfate)
Cesium: $\text{Cs}^+$, Sulfate: $\text{SO}_4^{2-}$
Step15: Balance charges (cesium sulfate)
$2\text{Cs}^+ + \text{SO}_4^{2-}
ightarrow \text{Cs}_2\text{SO}_4$
Step16: Identify ion charges (potassium dichromate)
Potassium: $\text{K}^+$, Dichromate: $\text{Cr}_2\text{O}_7^{2-}$
Step17: Balance charges (potassium dichromate)
$2\text{K}^+ + \text{Cr}_2\text{O}_7^{2-}
ightarrow \text{K}_2\text{Cr}_2\text{O}_7$
Step18: Identify ion charges (magnesium chlorite)
Magnesium: $\text{Mg}^{2+}$, Chlorite: $\text{ClO}_2^-$
Step19: Balance charges (magnesium chlorite)
$\text{Mg}^{2+} + 2\text{ClO}_2^-
ightarrow \text{Mg}(\text{ClO}_2)_2$
Step20: Identify ion charges (lithium oxalate)
Lithium: $\text{Li}^+$, Oxalate: $\text{C}_2\text{O}_4^{2-}$
Step21: Balance charges (lithium oxalate)
$2\text{Li}^+ + \text{C}_2\text{O}_4^{2-}
ightarrow \text{Li}_2\text{C}_2\text{O}_4$
Step22: Identify ion charges (sodium carbonate)
Sodium: $\text{Na}^+$, Carbonate: $\text{CO}_3^{2-}$
Step23: Balance charges (sodium carbonate)
$2\text{Na}^+ + \text{CO}_3^{2-}
ightarrow \text{Na}_2\text{CO}_3$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{AlPO}_3$
- $\text{K}_2\text{SO}_3$
- $\text{Al}_2(\text{CrO}_4)_3$
- $\text{LiNO}_3$
- $\text{Sr}(\text{ClO}_4)_2$
- $\text{Be}(\text{OH})_2$
- $\text{Ca}(\text{C}_2\text{H}_3\text{O}_2)_2$
- $\text{Cs}_2\text{SO}_4$
- $\text{K}_2\text{Cr}_2\text{O}_7$
- $\text{Mg}(\text{ClO}_2)_2$
- $\text{Li}_2\text{C}_2\text{O}_4$
- $\text{Na}_2\text{CO}_3$