QUESTION IMAGE
Question
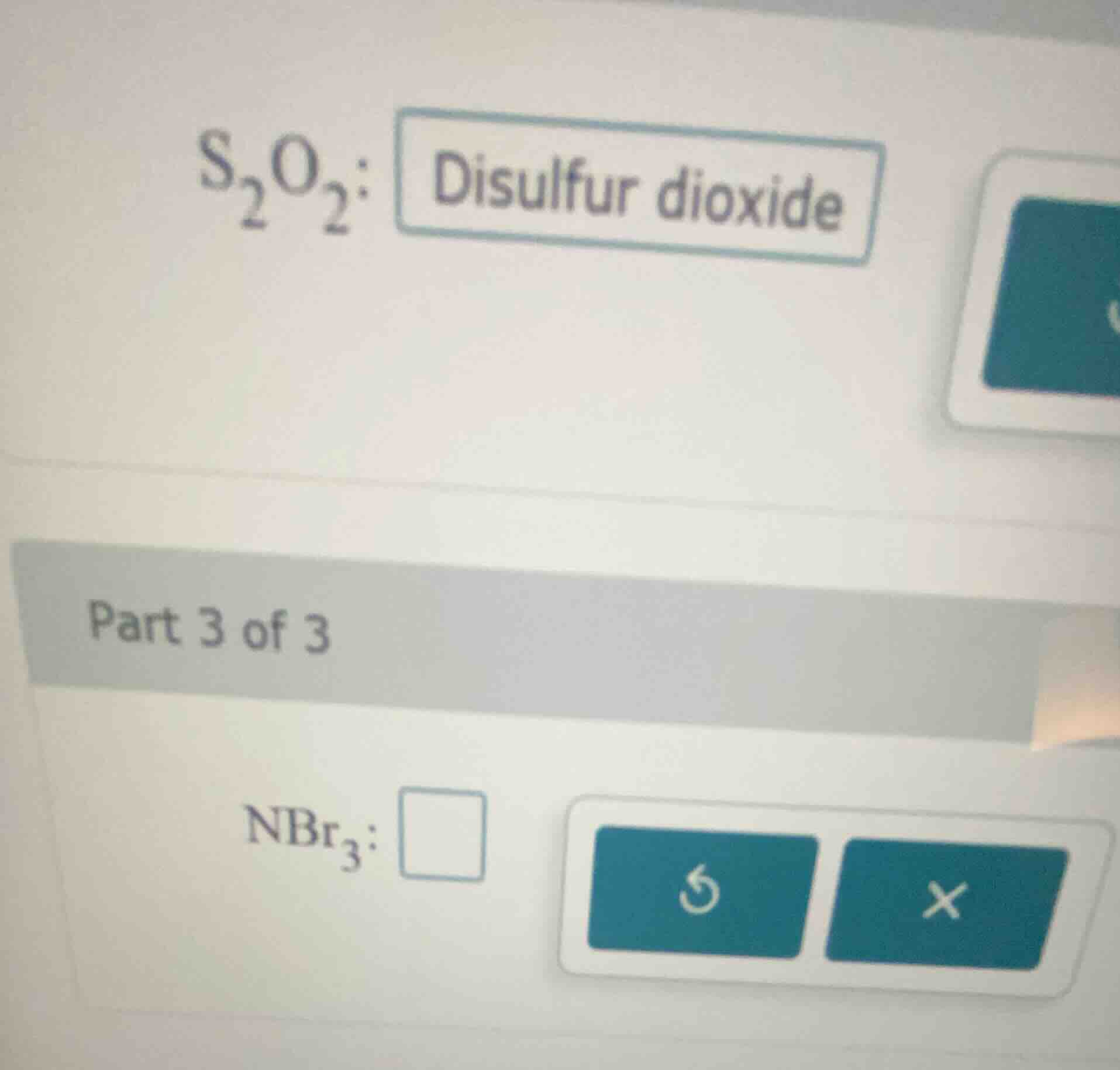
s₂o₂: disulfur dioxide
part 3 of 3
nbr₃:
Step1: Identify the elements and their prefixes
The compound is \( \text{NBr}_3 \). The first element is nitrogen (N), and the second is bromine (Br). For binary molecular compounds, we use prefixes based on the subscript. The subscript for N is 1 (so we use "mono-" but we usually omit "mono-" for the first element), and the subscript for Br is 3, so we use "tri-".
Step2: Name the compound
The name of the first element (nitrogen) remains as is, and the second element (bromine) changes its ending to -ide. So with the prefix "tri-" for bromine, the name is Nitrogen tribromide.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Nitrogen tribromide