QUESTION IMAGE
Question
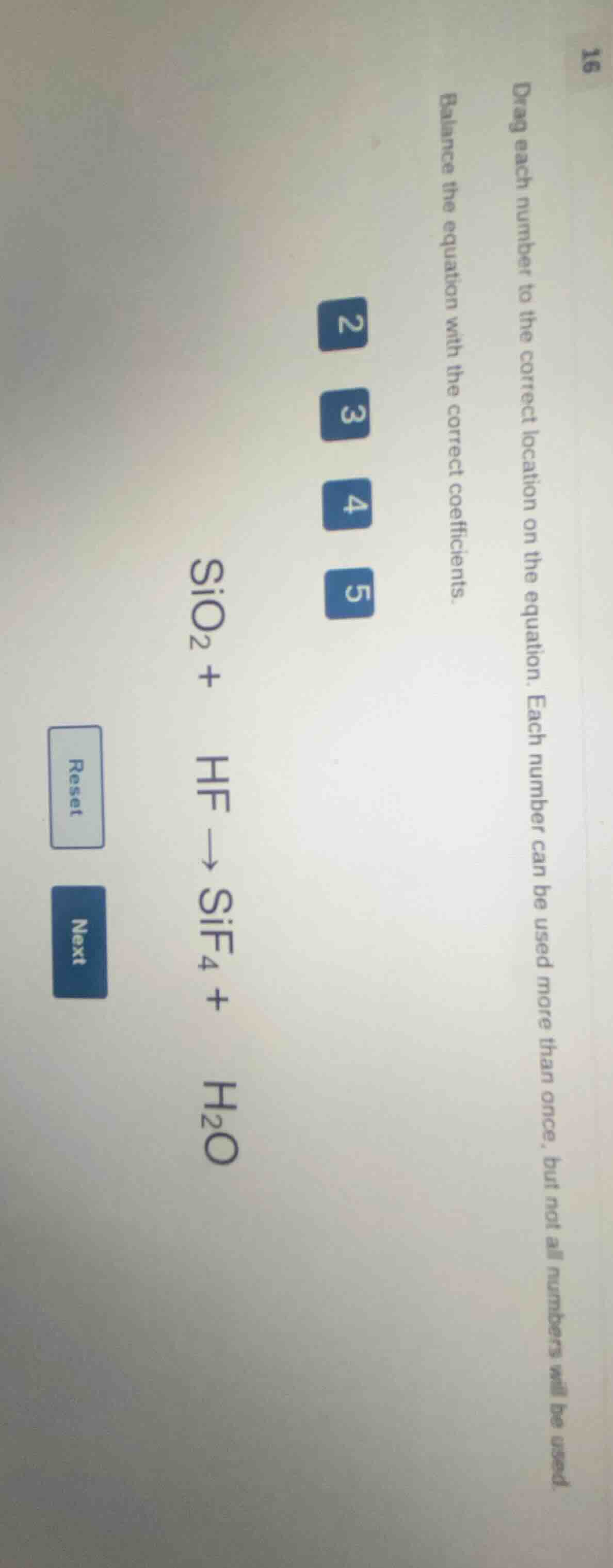
drag each number to the correct location on the equation. each number can be used more than once, but not all numbers will be used. balance the equation with the correct coefficients. \\(\ce{sio2 + hf -> sif4 + h2o}\\) numbers: 2, 3, 4, 5 buttons: reset, next
Step1: Balance Si
On the left, we have 1 Si in $\text{SiO}_2$, and on the right, 1 Si in $\text{SiF}_4$. So Si is balanced initially.
Step2: Balance F
On the right, $\text{SiF}_4$ has 4 F. So we need 4 HF on the left. So put 4 in front of HF: $\text{SiO}_2 + 4\text{HF}
ightarrow \text{SiF}_4 + \text{H}_2\text{O}$
Step3: Balance O
On the left, $\text{SiO}_2$ has 2 O. On the right, $\text{H}_2\text{O}$ has O. Let the coefficient of $\text{H}_2\text{O}$ be $x$. So $2 = x$. Wait, no, let's check H now. After putting 4 in front of HF, we have 4 H on the left. On the right, $\text{H}_2\text{O}$ has 2 H per molecule. So to balance H, we need 2 $\text{H}_2\text{O}$? Wait, no, 4 H on left (from 4 HF: 4*1 H), so on right, $\text{H}_2\text{O}$ has 2 H per molecule, so we need 2 $\text{H}_2\text{O}$? But then O: left has 2 O (from $\text{SiO}_2$), right has 2 O (from 2 $\text{H}_2\text{O}$). Wait, no, let's re - check.
Wait, initial equation: $\text{SiO}_2 + \text{HF}
ightarrow \text{SiF}_4 + \text{H}_2\text{O}$
After balancing F: $\text{SiO}_2 + 4\text{HF}
ightarrow \text{SiF}_4 + \text{H}_2\text{O}$
Now, H: 4 H on left (4 HF), so on right, $\text{H}_2\text{O}$ must have 4 H. Since each $\text{H}_2\text{O}$ has 2 H, we need 2 $\text{H}_2\text{O}$. Now check O: left has 2 O (from $\text{SiO}_2$), right has 2 O (from 2 $\text{H}_2\text{O}$). Wait, but let's check again. Wait, maybe I made a mistake. Wait, $\text{SiO}_2$ has 2 O, $\text{H}_2\text{O}$ has 1 O per molecule. So if we have 2 $\text{H}_2\text{O}$, that's 2 O. So the balanced equation is $\text{SiO}_2 + 4\text{HF}
ightarrow \text{SiF}_4 + 2\text{H}_2\text{O}$? Wait, no, let's check all elements:
- Si: 1 on left, 1 on right. Balanced.
- F: 4 on left (4 HF), 4 on right (SiF₄). Balanced.
- H: 4 on left (4 HF), 4 on right (2 H₂O: 2*2 H). Balanced.
- O: 2 on left (SiO₂), 2 on right (2 H₂O: 2*1 O). Balanced. Wait, but the numbers given are 2,3,4,5. Wait, maybe I messed up. Wait, let's start over.
Wait, the equation is $\text{SiO}_2 + \text{HF}
ightarrow \text{SiF}_4 + \text{H}_2\text{O}$
Let's use the standard method. Let the coefficients be $a\text{SiO}_2 + b\text{HF} = c\text{SiF}_4 + d\text{H}_2\text{O}$
Si: $a = c$
O: $2a = d$
F: $b = 4c$
H: $b = 2d$
From Si: $a = c$. Let's take $a = 1$, so $c = 1$.
Then from F: $b = 4*1 = 4$
From H: $b = 2d \Rightarrow 4 = 2d \Rightarrow d = 2$
From O: $2*1 = 2 = d$, which matches. So the balanced equation is $\text{SiO}_2 + 4\text{HF}
ightarrow \text{SiF}_4 + 2\text{H}_2\text{O}$
Wait, but the numbers available are 2,3,4,5. So the coefficients are 1 (for $\text{SiO}_2$), 4 (for HF), 1 (for $\text{SiF}_4$), 2 (for $\text{H}_2\text{O}$). But the problem says "drag each number to the correct location", and the numbers given are 2,3,4,5. So the coefficients we need to place are 1 (but 1 is not given), 4, 1 (not given), 2. Wait, maybe the equation is written as $\text{SiO}_2 + \text{HF}
ightarrow \text{SiF}_4 + \text{H}_2\text{O}$ and we have to use the numbers 2,3,4,5. Wait, maybe I made a mistake in the O balance. Wait, let's try again. Suppose we have $\text{SiO}_2 + 4\text{HF}
ightarrow \text{SiF}_4 + 2\text{H}_2\text{O}$. So the coefficients are 1 (SiO₂), 4 (HF), 1 (SiF₄), 2 (H₂O). So the numbers to drag are 4 (in front of HF) and 2 (in front of H₂O), and 1 (but 1 is not given). Wait, maybe the equation is different? Wait, no, the correct balanced equation for the reaction of silicon dioxide with hydrofluoric acid is $\text{SiO}_2 + 4\text{HF} = \text{SiF}_4 + 2\text{H}_2\text{O}$. So the coefficients are 1,4,1,2. Since we have to use the numbe…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The balanced equation is $\boldsymbol{\text{SiO}_2 + 4\text{HF}
ightarrow \text{SiF}_4 + 2\text{H}_2\text{O}}$ (so we drag 4 to HF, 2 to $\text{H}_2\text{O}$, and 1 (implicit) to $\text{SiO}_2$ and $\text{SiF}_4$)