QUESTION IMAGE
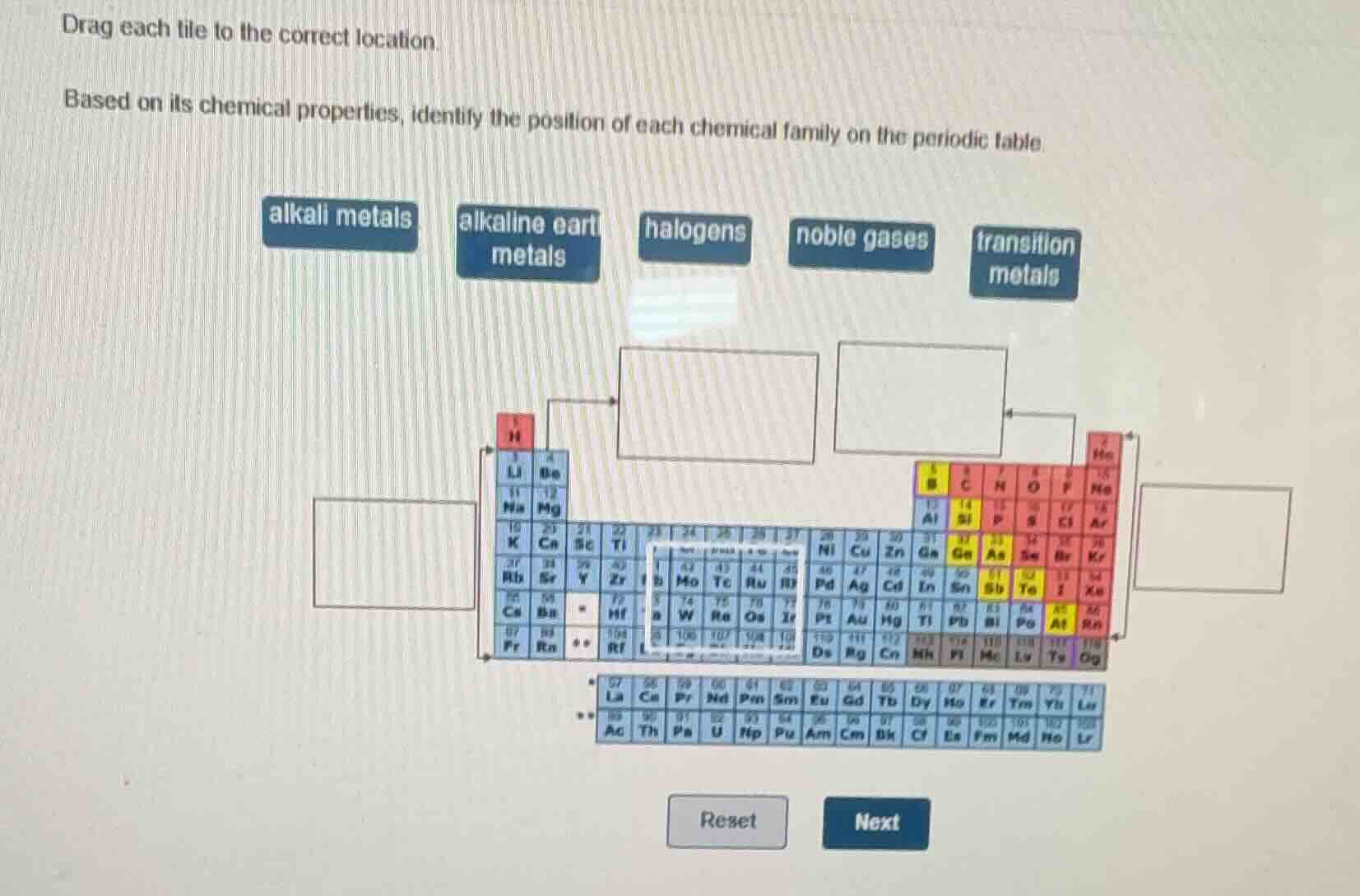
Question
drag each tile to the correct location.
based on its chemical properties, identify the position of each chemical family on the periodic table.
alkali metals
alkaline earth metals
halogens
noble gases
transition metals
reset
next
Brief Explanations
- Alkali metals are Group 1 (excluding hydrogen) of the periodic table, matching the leftmost blue column (Li, Na, K, Rb, Cs, Fr).
- Alkaline earth metals are Group 2, the second blue column (Be, Mg, Ca, Sr, Ba, Ra).
- Halogens are Group 17, the second-to-last red column (F, Cl, Br, I, At, Ts).
- Noble gases are Group 18, the rightmost red column (He, Ne, Ar, Kr, Xe, Rn, Og).
- Transition metals occupy the large central block of the periodic table, matching the white outlined section (groups 3-12).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Left empty box (Group 1, below H): alkali metals
- Top-left empty box (Group 2): alkaline earth metals
- Central empty box (Groups 3-12): transition metals
- Top-right empty box (Group 18): noble gases
- Right empty box (Group 17): halogens