QUESTION IMAGE
Question
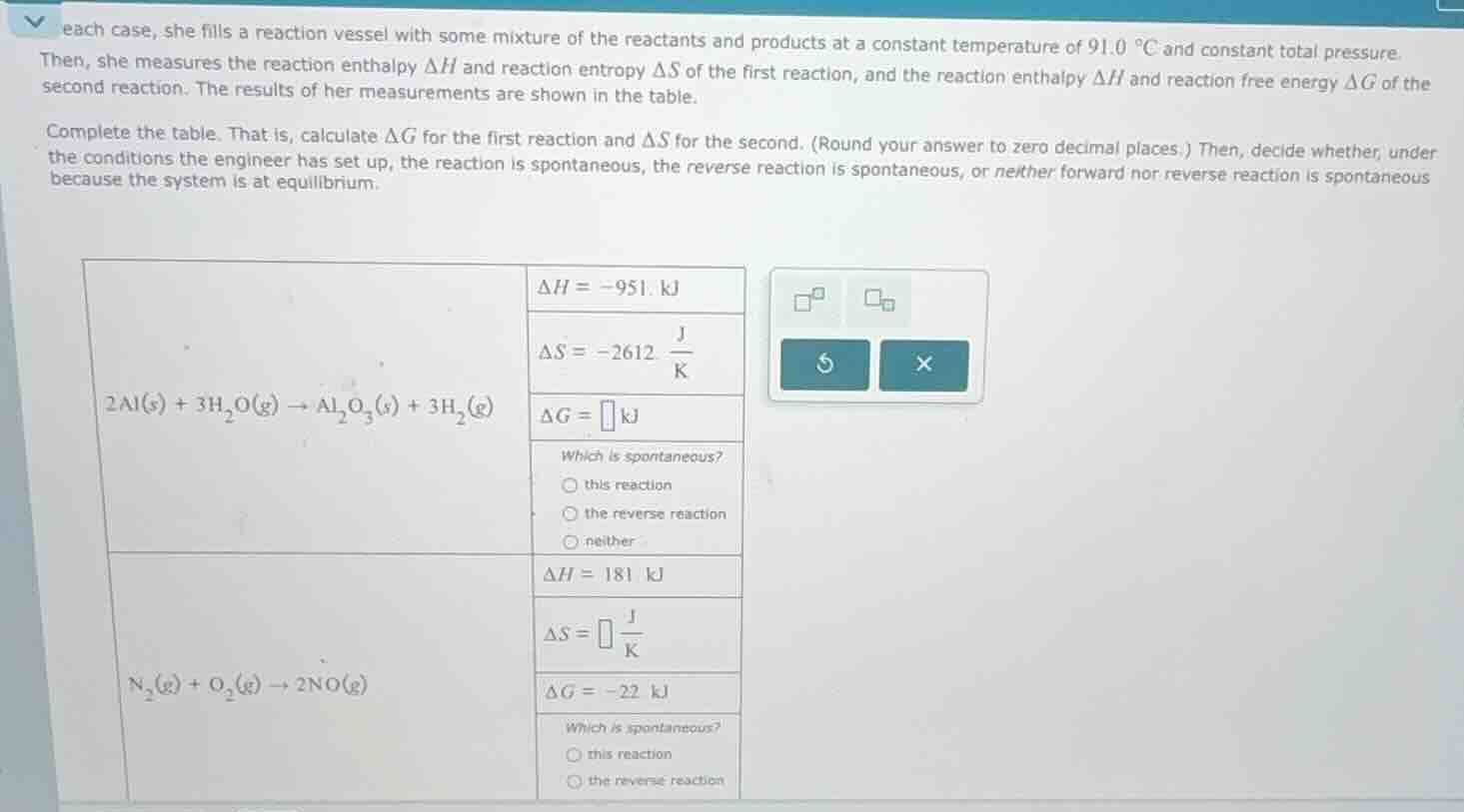
each case, she fills a reaction vessel with some mixture of the reactants and products at a constant temperature of 91.0 °c and constant total pressure. then, she measures the reaction enthalpy δh and reaction entropy δs of the first reaction, and the reaction enthalpy δh and reaction free energy δg of the second reaction. the results of her measurements are shown in the table. complete the table. that is, calculate δg for the first reaction and δs for the second. (round your answer to zero decimal places.) then, decide whether, under the conditions the engineer has set up, the reaction is spontaneous, the reverse reaction is spontaneous, or neither forward nor reverse reaction is spontaneous because the system is at equilibrium. \\(2\text{al}(s) + 3\text{h}_2\text{o}(g) \
ightarrow \text{al}_2\text{o}_3(s) + 3\text{h}_2(g)\\) \\(\delta h = -951\text{ kj}\\) \\(\delta s = -2612 \frac{\text{j}}{\text{k}}\\) \\(\delta g = \square \text{ kj}\\) which is spontaneous? \\(\circ\\) this reaction \\(\circ\\) the reverse reaction \\(\circ\\) neither \\(\text{n}_2(g) + \text{o}_2(g) \
ightarrow 2\text{no}(g)\\) \\(\delta h = 181\text{ kj}\\) \\(\delta s = \square \frac{\text{j}}{\text{k}}\\) \\(\delta g = -22\text{ kj}\\) which is spontaneous? \\(\circ\\) this reaction \\(\circ\\) the reverse reaction
First Reaction: \( \boldsymbol{2\text{Al}(s) + 3\text{H}_2\text{O}(g)
ightarrow \text{Al}_2\text{O}_3(s) + 3\text{H}_2(g)} \)
Step 1: Convert temperature to Kelvin
The temperature is \( 91.0^\circ \text{C} \). To convert to Kelvin, use \( T = 91.0 + 273.15 = 364.15 \, \text{K} \).
Step 2: Recall the Gibbs free energy formula
The formula for Gibbs free energy is \( \Delta G = \Delta H - T\Delta S \).
Step 3: Substitute the values
Given \( \Delta H = -951 \, \text{kJ} = -951000 \, \text{J} \), \( \Delta S = -2612 \, \frac{\text{J}}{\text{K}} \), and \( T = 364.15 \, \text{K} \).
First, calculate \( T\Delta S \):
\( T\Delta S = 364.15 \, \text{K} \times (-2612 \, \frac{\text{J}}{\text{K}}) = -364.15 \times 2612 \, \text{J} \approx -951159.8 \, \text{J} \approx -951.16 \, \text{kJ} \)
Now, calculate \( \Delta G \):
\( \Delta G = \Delta H - T\Delta S = -951000 \, \text{J} - (-951159.8 \, \text{J}) = -951000 + 951159.8 = 159.8 \, \text{J} \approx 0 \, \text{kJ} \) (rounded to zero decimal places). Wait, let's check the calculation again. Wait, \( \Delta H \) is in kJ, \( \Delta S \) is in J/K. Let's do unit conversion properly.
\( \Delta H = -951 \, \text{kJ} = -951000 \, \text{J} \)
\( T\Delta S = 364.15 \, \text{K} \times (-2612 \, \text{J/K}) = -364.15 \times 2612 = -951159.8 \, \text{J} = -951.16 \, \text{kJ} \)
Then \( \Delta G = \Delta H - T\Delta S = -951 \, \text{kJ} - (-951.16 \, \text{kJ}) = -951 + 951.16 = 0.16 \, \text{kJ} \approx 0 \, \text{kJ} \) (rounded to zero decimal places). Wait, maybe I made a mistake. Wait, \( \Delta H \) is -951 kJ, \( \Delta S \) is -2612 J/K. Let's compute \( T\Delta S \) in kJ:
\( T = 91 + 273.15 = 364.15 \, \text{K} \)
\( T\Delta S = 364.15 \, \text{K} \times (-2612 \, \text{J/K}) = -364.15 \times 2612 \, \text{J} = -951159.8 \, \text{J} = -951.16 \, \text{kJ} \)
Then \( \Delta G = \Delta H - T\Delta S = -951 \, \text{kJ} - (-951.16 \, \text{kJ}) = 0.16 \, \text{kJ} \approx 0 \, \text{kJ} \) (rounded to zero decimal places). So \( \Delta G \approx 0 \, \text{kJ} \).
Step 4: Determine spontaneity
If \( \Delta G = 0 \), the system is at equilibrium, so neither the forward nor reverse reaction is spontaneous.
Second Reaction: \( \boldsymbol{\text{N}_2(g) + \text{O}_2(g)
ightarrow 2\text{NO}(g)} \)
Step 1: Recall the Gibbs free energy formula
Again, use \( \Delta G = \Delta H - T\Delta S \). We need to solve for \( \Delta S \).
Step 2: Rearrange the formula
\( \Delta S = \frac{\Delta H - \Delta G}{T} \)
Step 3: Substitute the values
Given \( \Delta H = 181 \, \text{kJ} = 181000 \, \text{J} \), \( \Delta G = -22 \, \text{kJ} = -22000 \, \text{J} \), and \( T = 364.15 \, \text{K} \).
First, calculate \( \Delta H - \Delta G \):
\( \Delta H - \Delta G = 181000 \, \text{J} - (-22000 \, \text{J}) = 181000 + 22000 = 203000 \, \text{J} \)
Then, calculate \( \Delta S \):
\( \Delta S = \frac{203000 \, \text{J}}{364.15 \, \text{K}} \approx 557.46 \, \frac{\text{J}}{\text{K}} \approx 557 \, \frac{\text{J}}{\text{K}} \) (rounded to zero decimal places).
Step 4: Determine spontaneity
Given \( \Delta G = -22 \, \text{kJ} < 0 \), the forward reaction (this reaction) is spontaneous.
Final Answers
First Reaction:
- \( \Delta G = \boxed{0} \, \text{kJ} \)
- Spontaneity: neither
Second Reaction:
- \( \Delta S = \boxed{557} \, \frac{\text{J}}{\text{K}} \)
- Spontaneity: this reaction
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First Reaction: \( \boldsymbol{2\text{Al}(s) + 3\text{H}_2\text{O}(g)
ightarrow \text{Al}_2\text{O}_3(s) + 3\text{H}_2(g)} \)
Step 1: Convert temperature to Kelvin
The temperature is \( 91.0^\circ \text{C} \). To convert to Kelvin, use \( T = 91.0 + 273.15 = 364.15 \, \text{K} \).
Step 2: Recall the Gibbs free energy formula
The formula for Gibbs free energy is \( \Delta G = \Delta H - T\Delta S \).
Step 3: Substitute the values
Given \( \Delta H = -951 \, \text{kJ} = -951000 \, \text{J} \), \( \Delta S = -2612 \, \frac{\text{J}}{\text{K}} \), and \( T = 364.15 \, \text{K} \).
First, calculate \( T\Delta S \):
\( T\Delta S = 364.15 \, \text{K} \times (-2612 \, \frac{\text{J}}{\text{K}}) = -364.15 \times 2612 \, \text{J} \approx -951159.8 \, \text{J} \approx -951.16 \, \text{kJ} \)
Now, calculate \( \Delta G \):
\( \Delta G = \Delta H - T\Delta S = -951000 \, \text{J} - (-951159.8 \, \text{J}) = -951000 + 951159.8 = 159.8 \, \text{J} \approx 0 \, \text{kJ} \) (rounded to zero decimal places). Wait, let's check the calculation again. Wait, \( \Delta H \) is in kJ, \( \Delta S \) is in J/K. Let's do unit conversion properly.
\( \Delta H = -951 \, \text{kJ} = -951000 \, \text{J} \)
\( T\Delta S = 364.15 \, \text{K} \times (-2612 \, \text{J/K}) = -364.15 \times 2612 = -951159.8 \, \text{J} = -951.16 \, \text{kJ} \)
Then \( \Delta G = \Delta H - T\Delta S = -951 \, \text{kJ} - (-951.16 \, \text{kJ}) = -951 + 951.16 = 0.16 \, \text{kJ} \approx 0 \, \text{kJ} \) (rounded to zero decimal places). Wait, maybe I made a mistake. Wait, \( \Delta H \) is -951 kJ, \( \Delta S \) is -2612 J/K. Let's compute \( T\Delta S \) in kJ:
\( T = 91 + 273.15 = 364.15 \, \text{K} \)
\( T\Delta S = 364.15 \, \text{K} \times (-2612 \, \text{J/K}) = -364.15 \times 2612 \, \text{J} = -951159.8 \, \text{J} = -951.16 \, \text{kJ} \)
Then \( \Delta G = \Delta H - T\Delta S = -951 \, \text{kJ} - (-951.16 \, \text{kJ}) = 0.16 \, \text{kJ} \approx 0 \, \text{kJ} \) (rounded to zero decimal places). So \( \Delta G \approx 0 \, \text{kJ} \).
Step 4: Determine spontaneity
If \( \Delta G = 0 \), the system is at equilibrium, so neither the forward nor reverse reaction is spontaneous.
Second Reaction: \( \boldsymbol{\text{N}_2(g) + \text{O}_2(g)
ightarrow 2\text{NO}(g)} \)
Step 1: Recall the Gibbs free energy formula
Again, use \( \Delta G = \Delta H - T\Delta S \). We need to solve for \( \Delta S \).
Step 2: Rearrange the formula
\( \Delta S = \frac{\Delta H - \Delta G}{T} \)
Step 3: Substitute the values
Given \( \Delta H = 181 \, \text{kJ} = 181000 \, \text{J} \), \( \Delta G = -22 \, \text{kJ} = -22000 \, \text{J} \), and \( T = 364.15 \, \text{K} \).
First, calculate \( \Delta H - \Delta G \):
\( \Delta H - \Delta G = 181000 \, \text{J} - (-22000 \, \text{J}) = 181000 + 22000 = 203000 \, \text{J} \)
Then, calculate \( \Delta S \):
\( \Delta S = \frac{203000 \, \text{J}}{364.15 \, \text{K}} \approx 557.46 \, \frac{\text{J}}{\text{K}} \approx 557 \, \frac{\text{J}}{\text{K}} \) (rounded to zero decimal places).
Step 4: Determine spontaneity
Given \( \Delta G = -22 \, \text{kJ} < 0 \), the forward reaction (this reaction) is spontaneous.
Final Answers
First Reaction:
- \( \Delta G = \boxed{0} \, \text{kJ} \)
- Spontaneity: neither
Second Reaction:
- \( \Delta S = \boxed{557} \, \frac{\text{J}}{\text{K}} \)
- Spontaneity: this reaction