QUESTION IMAGE
Question
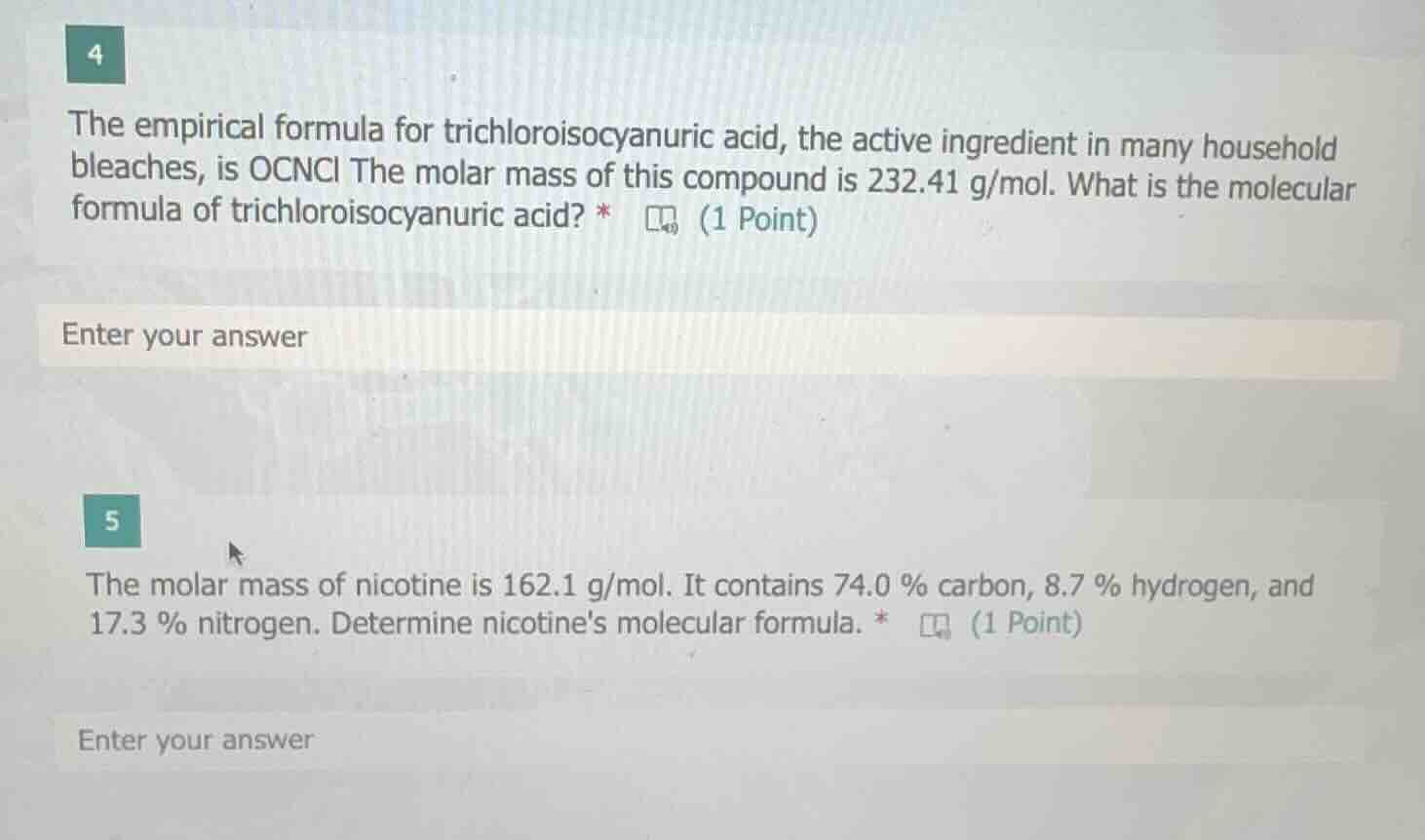
4
the empirical formula for trichloroisocyanuric acid, the active ingredient in many household bleaches, is ocncl the molar mass of this compound is 232.41 g/mol. what is the molecular formula of trichloroisocyanuric acid? *
enter your answer
5
the molar mass of nicotine is 162.1 g/mol. it contains 74.0 % carbon, 8.7 % hydrogen, and 17.3 % nitrogen. determine nicotines molecular formula. *
enter your answer
Question 4 Solution:
Step 1: Calculate empirical formula mass
The empirical formula is \( \text{OCNCl} \).
Atomic masses: \( \text{O} = 16.00 \, \text{g/mol} \), \( \text{C} = 12.01 \, \text{g/mol} \), \( \text{N} = 14.01 \, \text{g/mol} \), \( \text{Cl} = 35.45 \, \text{g/mol} \).
Empirical formula mass \( = 16.00 + 12.01 + 14.01 + 35.45 = 77.47 \, \text{g/mol} \).
Step 2: Find the ratio \( n \)
\( n = \frac{\text{Molar mass}}{\text{Empirical formula mass}} = \frac{232.41}{77.47} \approx 3 \).
Step 3: Determine molecular formula
Multiply each subscript in the empirical formula by \( n = 3 \).
Empirical formula: \( \text{OCNCl} \)
Molecular formula: \( \text{O}_3\text{C}_3\text{N}_3\text{Cl}_3 \) (or \( \text{C}_3\text{N}_3\text{O}_3\text{Cl}_3 \))
Question 5 Solution:
Step 1: Assume 100 g of sample
Mass of each element: \( \text{C} = 74.0 \, \text{g} \), \( \text{H} = 8.7 \, \text{g} \), \( \text{N} = 17.3 \, \text{g} \).
Step 2: Calculate moles of each element
Moles of \( \text{C} = \frac{74.0}{12.01} \approx 6.16 \, \text{mol} \)
Moles of \( \text{H} = \frac{8.7}{1.008} \approx 8.63 \, \text{mol} \)
Moles of \( \text{N} = \frac{17.3}{14.01} \approx 1.23 \, \text{mol} \)
Step 3: Find the simplest ratio (empirical formula)
Divide by the smallest number of moles (1.23):
\( \text{C}: \frac{6.16}{1.23} \approx 5 \), \( \text{H}: \frac{8.63}{1.23} \approx 7 \), \( \text{N}: \frac{1.23}{1.23} = 1 \).
Empirical formula: \( \text{C}_5\text{H}_7\text{N} \).
Step 4: Calculate empirical formula mass
Empirical formula mass \( = (5 \times 12.01) + (7 \times 1.008) + 14.01 = 60.05 + 7.056 + 14.01 = 81.116 \, \text{g/mol} \).
Step 5: Find \( n \)
\( n = \frac{162.1}{81.116} \approx 2 \).
Step 6: Determine molecular formula
Multiply subscripts by \( n = 2 \):
Molecular formula: \( \text{C}_{10}\text{H}_{14}\text{N}_2 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
Question 4: \( \boldsymbol{\text{C}_3\text{N}_3\text{O}_3\text{Cl}_3} \) (or \( \text{O}_3\text{C}_3\text{N}_3\text{Cl}_3 \))
Question 5: \( \boldsymbol{\text{C}_{10}\text{H}_{14}\text{N}_2} \)