QUESTION IMAGE
Question
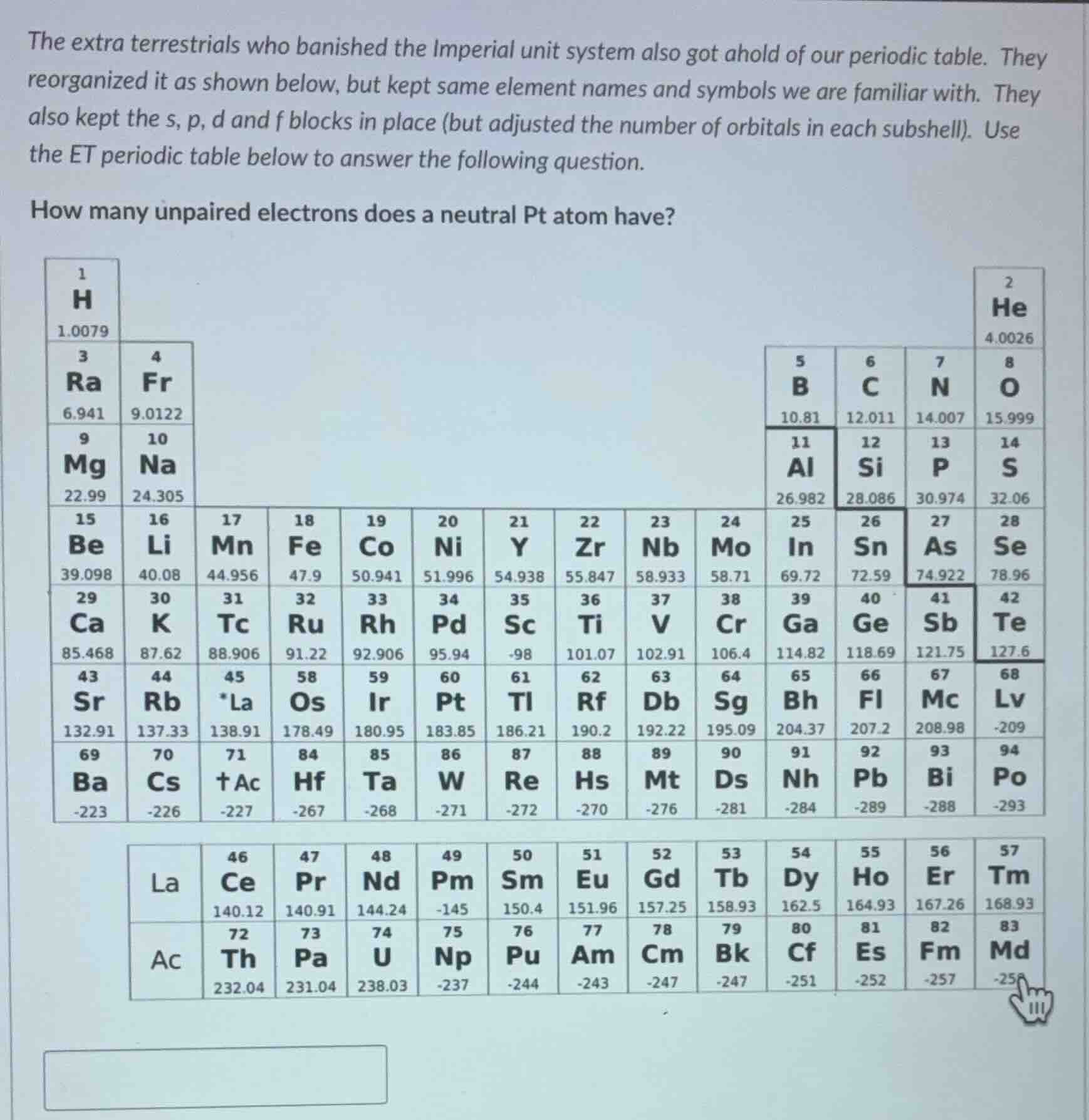
the extra terrestrials who banished the imperial unit system also got ahold of our periodic table. they reorganized it as shown below, but kept same element names and symbols we are familiar with. they also kept the s, p, d and f blocks in place (but adjusted the number of orbitals in each subshell). use the et periodic table below to answer the following question.
how many unpaired electrons does a neutral pt atom have?
Step1: Find Pt's atomic number
From the table, Pt (atomic number 60) is in the d-block (the middle block of the main table).
Step2: Define subshell orbital counts
The s, p, d, f blocks are kept in place, so we infer the block widths:
- s-block: 2 elements (groups 1,2) → s-subshell has 1 orbital (holds 2 e⁻)
- p-block: 4 elements per row (groups 5-8, 11-14, etc.) → p-subshell has 2 orbitals (holds 4 e⁻)
- d-block: 10 elements per row (positions 17-26, 31-40, etc.) → d-subshell has 5 orbitals (holds 10 e⁻)
Step3: Build electron configuration
Fill subshells in order of the table's row (period) filling:
- Period 1: $1s^2$ (2 e⁻)
- Period 2: $2s^22p^4$ (2+4=6 e⁻; total=8)
- Period 3: $3s^23p^4$ (2+4=6 e⁻; total=14)
- Period 4: $4s^23d^{10}4p^4$ (2+10+4=16 e⁻; total=30)
- Period 5: $5s^24d^{10}5p^4$ (2+10+4=16 e⁻; total=46)
- Period 6: Fill $6s^2$ (2 e⁻; total=48), then fill 5d subshell for Pt: $5d^{12}$? No, Pt is 12th element in period 6: $6s^25d^{12}$ is wrong. Wait, period 6 starts at Sr (43, Z=38? No, Pt is Z=60. Total e⁻=60. Subtract previous totals: 60-46=14. So $6s^25d^{12}$ invalid. Wait, d-block has 10 orbitals? No, 10 elements per d-block row: 17-26 is 10 elements (Mn to Sn), so d-subshell holds 10 e⁻. So period 6: $6s^2$, then 5d fills to 10, then 6p. Pt is 60-46=14 e⁻ into period 6: $6s^25d^{10}6p^2$. Now, 5d is full (10 e⁻, all paired), 6s is full (2 e⁻, paired), 6p has 2 e⁻. Since p-subshell has 2 orbitals, Hund's rule says each orbital gets one e⁻ first. So 6p has 2 unpaired e⁻.
Wait, correction: Pt is in position 60, count electrons:
Period1: 2, Period2:6, Period3:6, Period4:16, Period5:16. Total: 2+6+6+16+16=46. 60-46=14. Period6: first 2 are s-block (Sr, Rb: Z=38,39? No, Z=60 is Pt, which is 3rd element in the d-block of period6 (Os=58, Ir=59, Pt=60). So period6 fills $6s^2$ (Sr, Rb: Z=43,44, 2 e⁻), then 5d starts at Os (58, Z=58: 5d¹, Ir=59:5d², Pt=60:5d³? No, total e⁻=46+2+3=51, no. Wait, the f-block is separate, so period6 includes f-block (La, Ce...Tm, 14 elements). So total e⁻ up to end of f-block: 46+14=60. Oh! Pt is Z=60, which is the last element of the f-block? No, Pt is in the main table, f-block is below. So main table period6: Sr(43), Rb(44), La*(45), Os(58), Ir(59), Pt(60)... So f-block is 14 elements (Z=46-59? No, La is 45, Ce=46...Tm=57, 12 elements? No, 57-46+1=12. So total e⁻ up to end of f-block: 46+12=58. Then Os=58 (Z=58: 6s²5d¹), Ir=59:6s²5d², Pt=60:6s²5d³. Now, 5d subshell has 5 orbitals, so 3 e⁻ fill 3 separate orbitals (Hund's rule), all unpaired. Wait, no, s-subshell is 1 orbital, p is 2, d is 5. So d-subshell has 5 orbitals, each holds 2 e⁻. 3 e⁻ in 5d: 3 unpaired e⁻.
Wait, no, let's count Z correctly: H=1, He=2, Ra=3, Fr=4, B=5, C=6, N=7, O=8, Mg=9, Na=10, Al=11, Si=12, P=13, S=14, Be=15, Li=16, Mn=17, Fe=18, Co=19, Ni=20, Y=21, Zr=22, Nb=23, Mo=24, In=25, Sn=26, As=27, Se=28, Ca=29, K=30, Tc=31, Ru=32, Rh=33, Pd=34, Sc=35, Ti=36, V=37, Cr=38, Ga=39, Ge=40, Sb=41, Te=42, Sr=43, Rb=44, La*=45, Os=58, Ir=59, Pt=60... Yes, Pt is Z=60.
Now, electron filling order: follow the periodic table's block order: s (1st two columns), p (next four), d (next ten), f (bottom block).
- 1s² (H, He: 2 e⁻)
- 2s² (Ra, Fr: 2 e⁻; total=4), 2p⁴ (B, C, N, O:4 e⁻; total=8)
- 3s² (Mg, Na:2 e⁻; total=10), 3p⁴ (Al, Si, P, S:4 e⁻; total=14)
- 4s² (Be, Li:2 e⁻; total=16), 3d¹⁰ (Mn to Sn:10 e⁻; total=26), 4p⁴ (As, Se, In, Sn? No, In=25, Sn=26, As=27, Se=28: 4 e⁻; total=30)
- 5s² (Ca, K:2 e⁻; total=32), 4d¹⁰ (Tc to Ge:10 e⁻; total=42), 5p⁴ (Sb, Te, Ga, Ge? No, Ga=39, Ge=40, Sb=41, Te=42:4 e⁻; total=46)
- 6s² (Sr…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
3