QUESTION IMAGE
Question
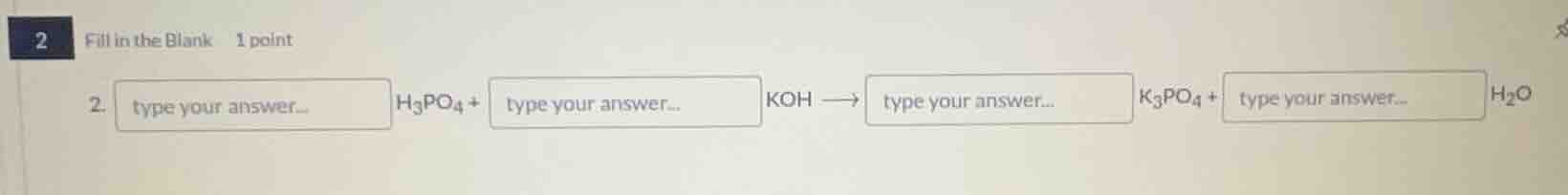
2 fill in the blank 1 point 2. type your answer... h₃po₄ + type your answer... koh → type your answer... k₃po₄ + type your answer... h₂o
Step1: Identify the reaction type
This is an acid - base (neutralization) reaction between phosphoric acid ($\ce{H3PO4}$) and potassium hydroxide ($\ce{KOH}$). The products are potassium phosphate ($\ce{K3PO4}$) and water ($\ce{H2O}$). We need to balance the chemical equation.
Step2: Balance the potassium atoms
On the product side, in $\ce{K3PO4}$, there are 3 K atoms. So we need 3 $\ce{KOH}$ on the reactant side. So the coefficient of $\ce{KOH}$ is 3.
Step3: Balance the hydrogen and oxygen atoms (for water)
Now, on the reactant side, from $\ce{H3PO4}$ we have 3 H atoms and from 3 $\ce{KOH}$ we have 3 H atoms, so total 6 H atoms. Since each $\ce{H2O}$ has 2 H atoms, the number of $\ce{H2O}$ molecules will be 3 (because $6\div2 = 3$). Also, for the phosphate group, $\ce{H3PO4}$ has 1 $\ce{PO4^{3 - }}$ group and $\ce{K3PO4}$ has 1 $\ce{PO4^{3 - }}$ group, so the coefficient of $\ce{H3PO4}$ is 1.
The balanced chemical equation is: $\ce{H3PO4 + 3KOH -> K3PO4 + 3H2O}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First blank: 1 (coefficient of $\ce{H3PO4}$)
Second blank: 3 (coefficient of $\ce{KOH}$)
Third blank: 1 (coefficient of $\ce{K3PO4}$)
Fourth blank: 3 (coefficient of $\ce{H2O}$)
So filling in the blanks:
1 $\ce{H3PO4}$ + 3 $\ce{KOH}$ $
ightarrow$ 1 $\ce{K3PO4}$ + 3 $\ce{H2O}$