QUESTION IMAGE
Question
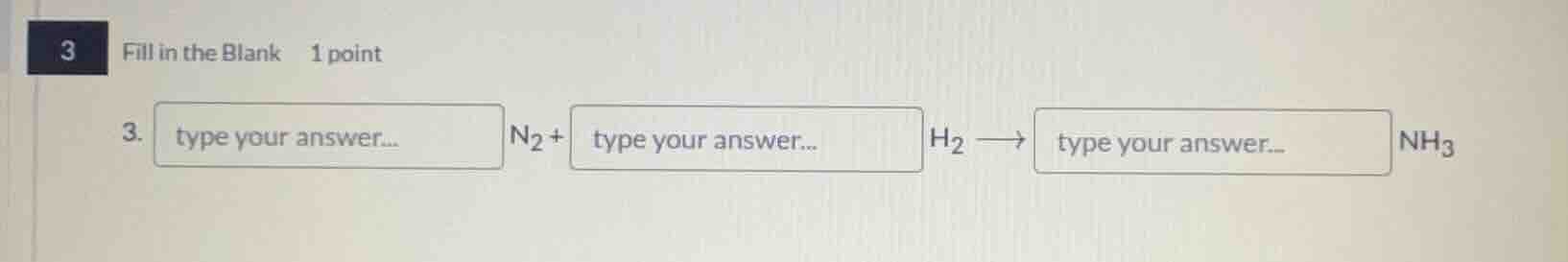
3 fill in the blank 1 point 3. type your answer... n₂ + type your answer... h₂ ⟶ type your answer... nh₃
Step1: Identify the reaction type
This is a chemical reaction for the synthesis of ammonia, known as the Haber process. The unbalanced equation is \( \text{N}_2 + \text{H}_2
ightarrow \text{NH}_3 \).
Step2: Balance the nitrogen atoms
There are 2 nitrogen atoms on the left (in \( \text{N}_2 \)) and 1 on the right (in \( \text{NH}_3 \)). So we put a coefficient of 2 in front of \( \text{NH}_3 \) to balance nitrogen: \( \text{N}_2 + \text{H}_2
ightarrow 2\text{NH}_3 \).
Step3: Balance the hydrogen atoms
Now, on the right, we have \( 2\times3 = 6 \) hydrogen atoms (from \( 2\text{NH}_3 \)). On the left, we have 2 hydrogen atoms (in \( \text{H}_2 \)). So we need a coefficient of 3 in front of \( \text{H}_2 \) to get 6 hydrogen atoms: \( \text{N}_2 + 3\text{H}_2
ightarrow 2\text{NH}_3 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The balanced equation is \( \boldsymbol{1}\text{N}_2 + \boldsymbol{3}\text{H}_2
ightarrow \boldsymbol{2}\text{NH}_3 \), so the coefficients (from left to right) are 1, 3, and 2.