QUESTION IMAGE
Question
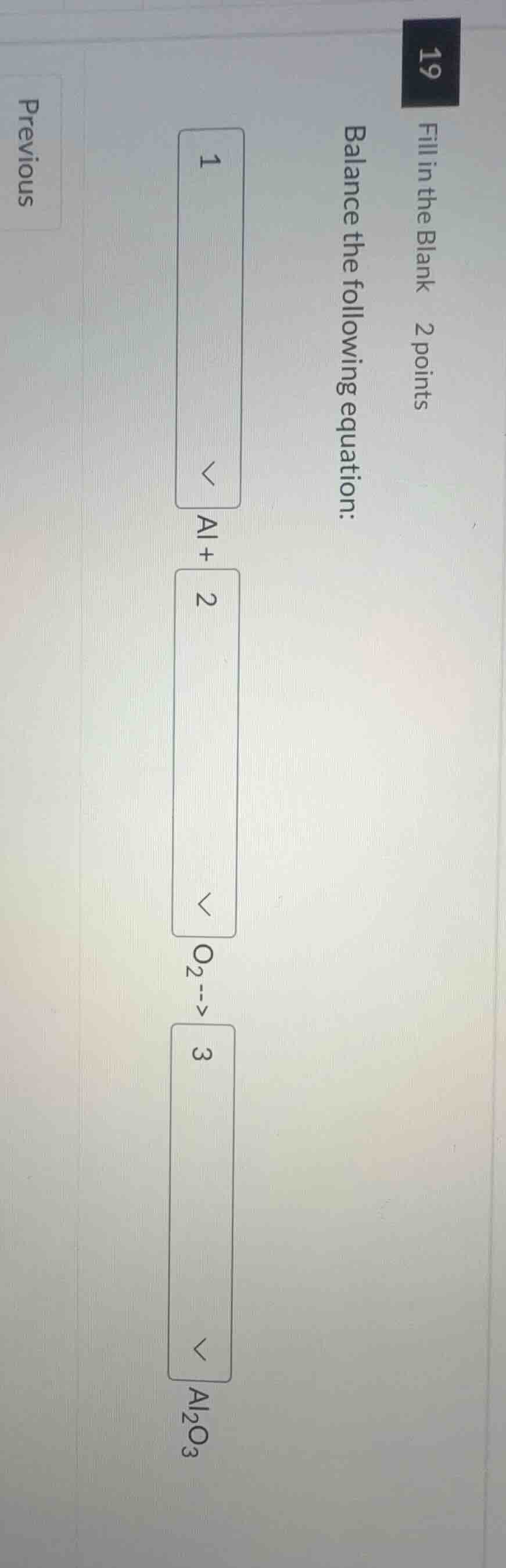
fill in the blank 2 points
balance the following equation:
1
al + 2
o₂ --> 3
al₂o₃
Step1: Identify atoms to balance
We need to balance the equation \( \text{Al} + \text{O}_2
ightarrow \text{Al}_2\text{O}_3 \). First, list the number of each atom on both sides. On the left: Al = 1, O = 2. On the right: Al = 2, O = 3.
Step2: Balance oxygen atoms
Find the least common multiple of 2 and 3, which is 6. So, for \( \text{O}_2 \), multiply by 3 (to get 6 O atoms: \( 3\times2 = 6 \)), and for \( \text{Al}_2\text{O}_3 \), multiply by 2 (to get 6 O atoms: \( 2\times3 = 6 \)). Now the equation is \( \text{Al} + 3\text{O}_2
ightarrow 2\text{Al}_2\text{O}_3 \).
Step3: Balance aluminum atoms
Now, on the right, Al atoms are \( 2\times2 = 4 \). So, multiply Al on the left by 4. The balanced equation is \( 4\text{Al} + 3\text{O}_2
ightarrow 2\text{Al}_2\text{O}_3 \).
Looking at the blanks:
- First blank (Al coefficient): 4
- Second blank (O₂ coefficient): 3
- Third blank (Al₂O₃ coefficient): 2 (but the problem shows the third blank as for Al₂O₃, and in the given structure, let's check the positions. Wait, the original blanks: first is Al, second is O₂, third is Al₂O₃. So:
First blank (Al): 4
Second blank (O₂): 3
Third blank (Al₂O₃): 2
Wait, but the user's image shows the blanks as:
- Al (first blank)
- O₂ (second blank)
- Al₂O₃ (third blank)
So let's re-express the balancing:
Start with \( \text{Al} + \text{O}_2
ightarrow \text{Al}_2\text{O}_3 \)
Balance O:
Multiply \( \text{O}_2 \) by 3: \( 3\text{O}_2 \) (gives 6 O)
Multiply \( \text{Al}_2\text{O}_3 \) by 2: \( 2\text{Al}_2\text{O}_3 \) (gives 6 O and 4 Al)
Then balance Al: multiply Al by 4: \( 4\text{Al} \)
So the coefficients are:
4 for Al, 3 for O₂, 2 for Al₂O₃.
But the user's blanks:
First blank (Al): 4
Second blank (O₂): 3
Third blank (Al₂O₃): 2
Wait, but in the image, the third blank is labeled as Al₂O₃, so:
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First blank (Al): 4
Second blank (O₂): 3
Third blank (Al₂O₃): 2
(But let's check the problem's blank positions. The first blank is before Al, second before O₂, third before Al₂O₃. So:
- 4
- 3
- 2)