QUESTION IMAGE
Question
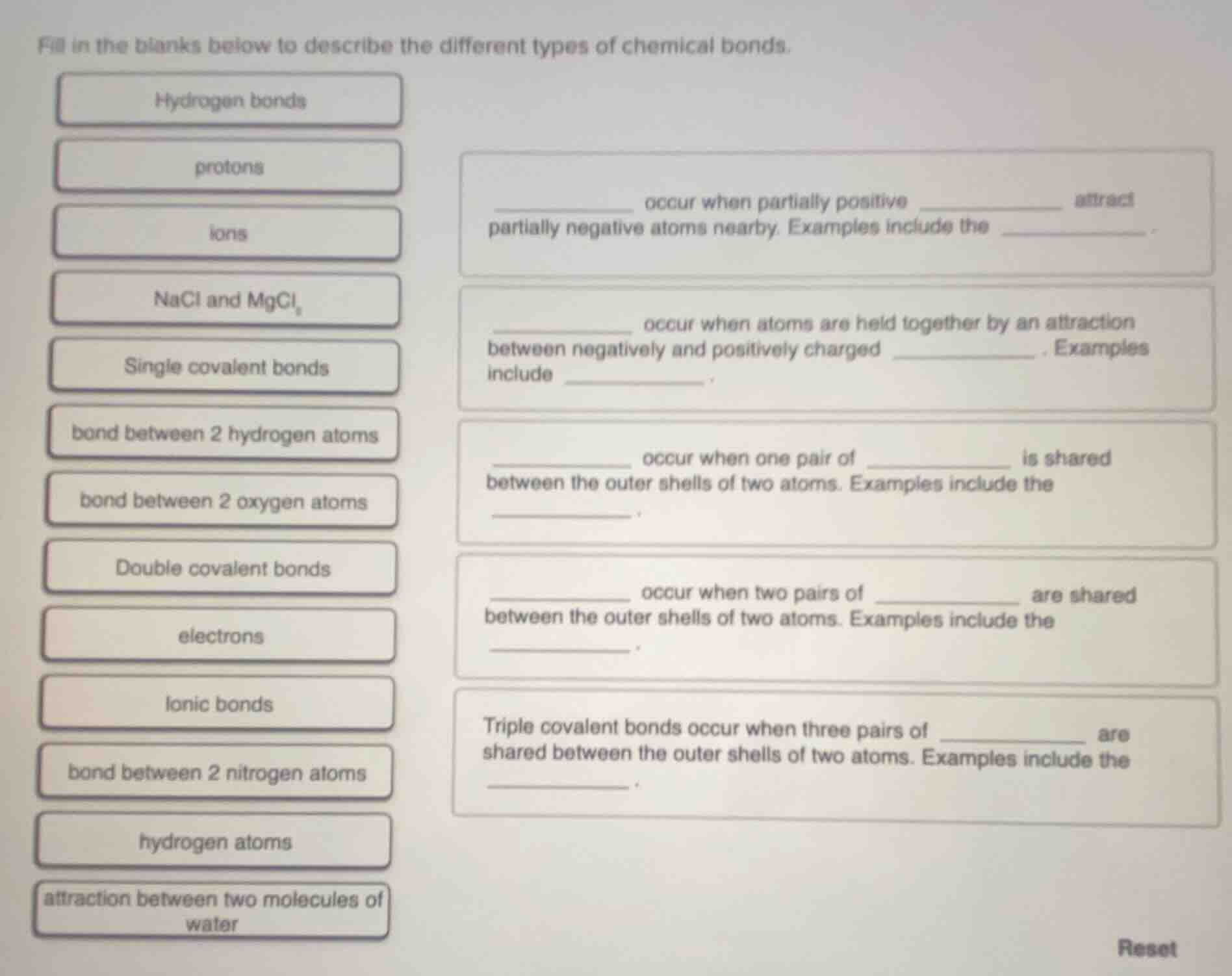
fill in the blanks below to describe the different types of chemical bonds.
hydrogen bonds
protons
ions
nacl and mgcl₂
single covalent bonds
bond between 2 hydrogen atoms
bond between 2 oxygen atoms
double covalent bonds
electrons
ionic bonds
bond between 2 nitrogen atoms
hydrogen atoms
attraction between two molecules of water
____ occur when partially positive __ attract partially negative atoms nearby. examples include the ____.
____ occur when atoms are held together by an attraction between negatively and positively charged __. examples include ____.
____ occur when one pair of __ is shared between the outer shells of two atoms. examples include the ____.
____ occur when two pairs of __ are shared between the outer shells of two atoms. examples include the ____.
triple covalent bonds occur when three pairs of ____ are shared between the outer shells of two atoms. examples include the ____.
First Blank Set:
Step1: Identify bond type for partial charges
Hydrogen bonds involve attraction between partially positive and negative atoms. So first blank: Hydrogen bonds.
Step2: Identify positive atom in H-bonds
In H-bonds, partially positive hydrogen atoms attract. So second blank: hydrogen atoms.
Step3: Example of H-bonds
Attraction between two water molecules is a hydrogen bond example. So third blank: attraction between two molecules of water.
Second Blank Set:
Step1: Bond with charged particles
Ionic bonds are between positive and negative ions. So first blank: Ionic bonds.
Step2: Charged particles in ionic bonds
Ionic bonds involve ions (charged atoms). So second blank: ions.
Step3: Ionic bond examples
NaCl and MgCl₂ are ionic compounds. So third blank: NaCl and MgCl₂.
Third Blank Set:
Step1: Bond with one electron pair
Single covalent bonds share one pair of electrons. So first blank: Single covalent bonds.
Step2: Particle shared in covalent bonds
Covalent bonds share electrons. So second blank: electrons.
Step3: Single covalent bond example
Bond between 2 hydrogen atoms is a single covalent bond. So third blank: bond between 2 hydrogen atoms.
Fourth Blank Set:
Step1: Bond with two electron pairs
Double covalent bonds share two pairs of electrons. So first blank: Double covalent bonds.
Step2: Particle shared in double covalent
Electrons are shared in double covalent bonds. So second blank: electrons.
Step3: Double covalent bond example
Bond between 2 oxygen atoms is a double covalent bond. So third blank: bond between 2 oxygen atoms.
Fifth Blank Set:
Step1: Particle shared in triple covalent
Triple covalent bonds share three pairs of electrons. So first blank: electrons.
Step2: Triple covalent bond example
Bond between 2 nitrogen atoms is a triple covalent bond. So second blank: bond between 2 nitrogen atoms.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Hydrogen bonds; hydrogen atoms; attraction between two molecules of water
- Ionic bonds; ions; NaCl and MgCl₂
- Single covalent bonds; electrons; bond between 2 hydrogen atoms
- Double covalent bonds; electrons; bond between 2 oxygen atoms
- electrons; bond between 2 nitrogen atoms