QUESTION IMAGE
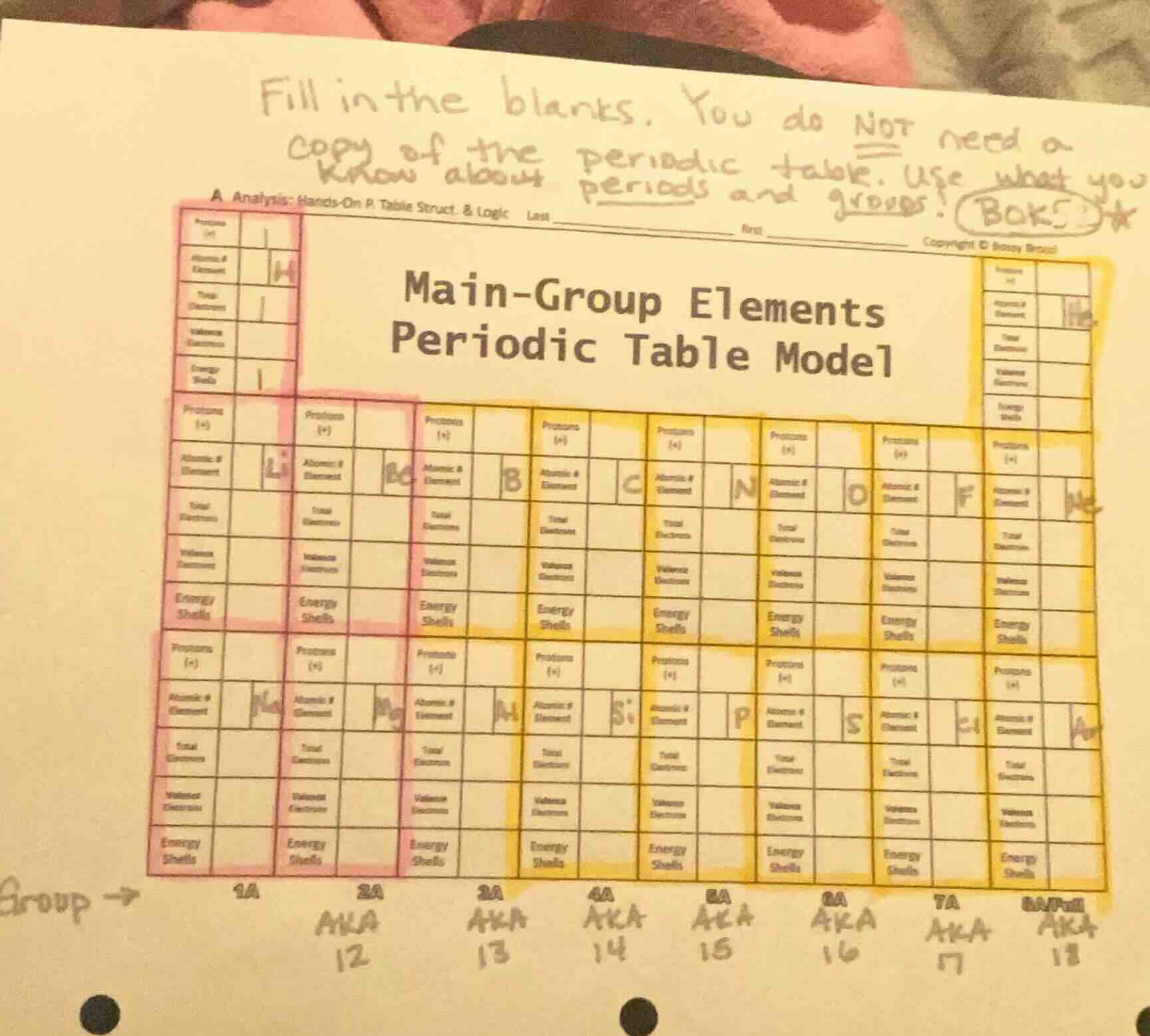
Question
fill in the blanks. you do not need a copy of the periodic table. use what you know about periods and groups!a analysis: hands-on p. table struct. & logic last _____ first _____ copyright © bossey broadmain-group elementsperiodic table modelgroup →1a 2a aka 12 3a aka 13 4a aka 14 5a aka 15 6a aka 16 7a aka 17 8a/0 aka 18
Step1: Define key rules
- Atomic number = number of protons = total electrons (neutral atoms)
- Energy shells = period number (row number)
- Valence electrons = group number (for main groups: 1A=1, 2A=2, 3A=3, 4A=4, 5A=5, 6A=6, 7A=7, 8A=8)
---
Row 1 (Period 1, Energy Shells=1)
Step2: Fill Hydrogen (H)
Protons = 1, Total Electrons = 1, Valence Electrons = 1
Step3: Fill Helium (He)
Protons = 2, Total Electrons = 2, Valence Electrons = 2 (special case for 8A, period 1)
---
Row 2 (Period 2, Energy Shells=2)
Step4: Fill Lithium (Li, Group 1A)
Protons = 3, Total Electrons = 3, Valence Electrons = 1
Step5: Fill Beryllium (Be, Group 2A)
Protons = 4, Total Electrons = 4, Valence Electrons = 2
Step6: Fill Boron (B, Group 3A)
Protons = 5, Total Electrons = 5, Valence Electrons = 3
Step7: Fill Carbon (C, Group 4A)
Protons = 6, Total Electrons = 6, Valence Electrons = 4
Step8: Fill Nitrogen (N, Group 5A)
Protons = 7, Total Electrons = 7, Valence Electrons = 5
Step9: Fill Oxygen (O, Group 6A)
Protons = 8, Total Electrons = 8, Valence Electrons = 6
Step10: Fill Fluorine (F, Group 7A)
Protons = 9, Total Electrons = 9, Valence Electrons = 7
Step11: Fill Neon (Ne, Group 8A)
Protons = 10, Total Electrons = 10, Valence Electrons = 8
---
Row 3 (Period 3, Energy Shells=3)
Step12: Fill Sodium (Na, Group 1A)
Protons = 11, Total Electrons = 11, Valence Electrons = 1
Step13: Fill Magnesium (Mg, Group 2A)
Protons = 12, Total Electrons = 12, Valence Electrons = 2
Step14: Fill Aluminum (Al, Group 3A)
Protons = 13, Total Electrons = 13, Valence Electrons = 3
Step15: Fill Silicon (Si, Group 4A)
Protons = 14, Total Electrons = 14, Valence Electrons = 4
Step16: Fill Phosphorus (P, Group 5A)
Protons = 15, Total Electrons = 15, Valence Electrons = 5
Step17: Fill Sulfur (S, Group 6A)
Protons = 16, Total Electrons = 16, Valence Electrons = 6
Step18: Fill Chlorine (Cl, Group 7A)
Protons = 17, Total Electrons = 17, Valence Electrons = 7
Step19: Fill Argon (Ar, Group 8A)
Protons = 18, Total Electrons = 18, Valence Electrons = 8
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Row 1 (Period 1)
| Property | H | He |
|---|---|---|
| Total Electrons | 1 | 2 |
| Valence Electrons | 1 | 2 |
| Energy Shells | 1 | 1 |
---
Row 2 (Period 2)
| Property | Li | Be | B | C | N | O | F | Ne |
|---|---|---|---|---|---|---|---|---|
| Total Electrons | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| Valence Electrons | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| Energy Shells | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
---
Row 3 (Period 3)
| Property | Na | Mg | Al | Si | P | S | Cl | Ar |
|---|---|---|---|---|---|---|---|---|
| Total Electrons | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
| Valence Electrons | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| Energy Shells | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 |