QUESTION IMAGE
Question
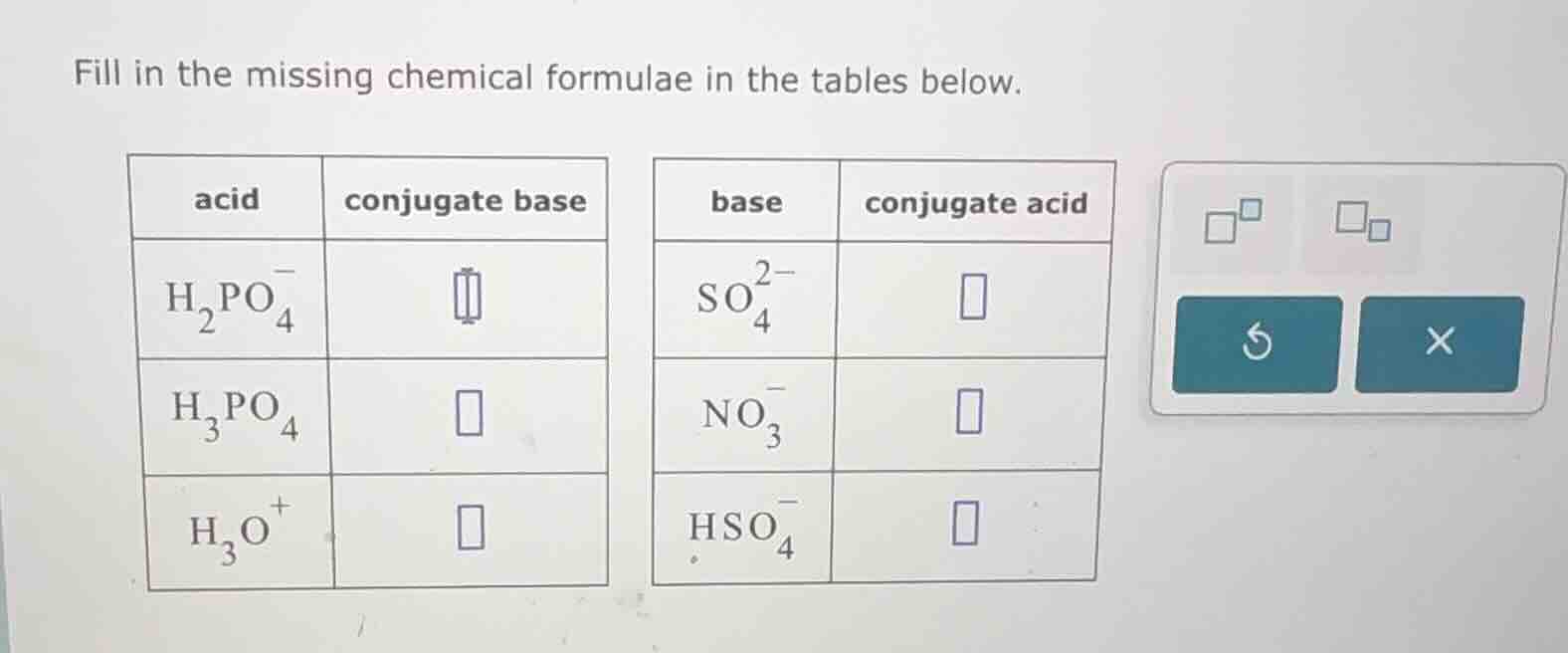
fill in the missing chemical formulae in the tables below.
acid\tconjugate base
base\tconjugate acid
$\ce{h_{2}po^{-}_{4}}$\t\t$\ce{so^{2-}_{4}}$\t
$\ce{h_{3}po_{4}}$\t\t$\ce{no^{-}_{3}}$\t
$\ce{h_{3}o^{+}}$\t\t$\ce{hso^{-}_{4}}$\t
To determine the conjugate base of an acid, the acid donates a proton ($\ce{H^+}$). To determine the conjugate acid of a base, the base accepts a proton ($\ce{H^+}$).
Step 1: Conjugate base of $\ce{H2PO4^-}$
$\ce{H2PO4^-}$ donates one $\ce{H^+}$:
$\ce{H2PO4^- -> HPO4^{2-} + H^+}$
So the conjugate base is $\ce{HPO4^{2-}}$.
Step 2: Conjugate base of $\ce{H3PO4}$
$\ce{H3PO4}$ donates one $\ce{H^+}$:
$\ce{H3PO4 -> H2PO4^- + H^+}$
So the conjugate base is $\ce{H2PO4^-}$.
Step 3: Conjugate base of $\ce{H3O^+}$
$\ce{H3O^+}$ donates one $\ce{H^+}$:
$\ce{H3O^+ -> H2O + H^+}$
So the conjugate base is $\ce{H2O}$.
Step 4: Conjugate acid of $\ce{SO4^{2-}}$
$\ce{SO4^{2-}}$ accepts one $\ce{H^+}$:
$\ce{SO4^{2-} + H^+ -> HSO4^-}$
So the conjugate acid is $\ce{HSO4^-}$.
Step 5: Conjugate acid of $\ce{NO3^-}$
$\ce{NO3^-}$ accepts one $\ce{H^+}$:
$\ce{NO3^- + H^+ -> HNO3}$
So the conjugate acid is $\ce{HNO3}$.
Step 6: Conjugate acid of $\ce{HSO4^-}$
$\ce{HSO4^-}$ accepts one $\ce{H^+}$:
$\ce{HSO4^- + H^+ -> H2SO4}$
So the conjugate acid is $\ce{H2SO4}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| acid | conjugate base | base | conjugate acid |
|---|---|---|---|
| $\ce{H3PO4}$ | $\ce{H2PO4^-}$ | $\ce{NO3^-}$ | $\ce{HNO3}$ |
| $\ce{H3O^+}$ | $\ce{H2O}$ | $\ce{HSO4^-}$ | $\ce{H2SO4}$ |