QUESTION IMAGE
Question
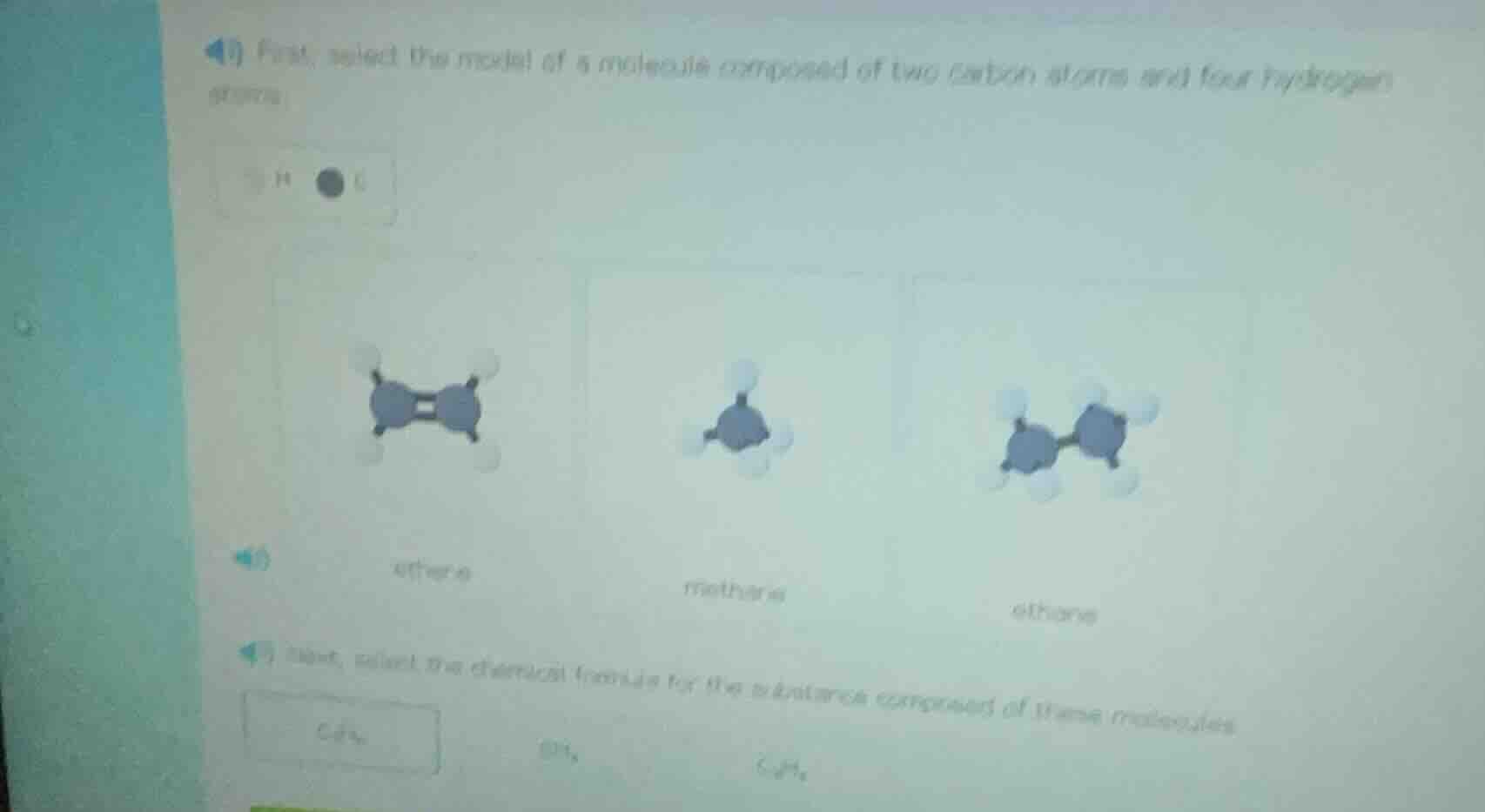
- first, select the model of a molecule composed of two carbon atoms and four hydrogen atoms.
then, select the chemical formula for the substance composed of these molecules.
Response
Part 1: Select the molecule model (Ethylene, Methane, Ethane)
Brief Explanations
- Methane: Has 1 carbon and 4 hydrogens ($CH_4$), so the middle model (1 C, 4 H) is methane.
- Ethane: Has 2 carbons and 6 hydrogens ($C_2H_6$), so the right model (2 C, 6 H) is ethane.
- Ethylene (Ethene): Has 2 carbons and 4 hydrogens ($C_2H_4$), with a double bond. The left model (2 C, 4 H, double bond) is ethylene.
The molecule with 2 C and 4 H is ethylene (ethene).
- Step 1: Ethylene’s formula: Ethylene (ethene) has the formula $C_2H_4$.
- Step 2: Multiply by 10: For 10 molecules, the formula is $10 \times C_2H_4 = C_{20}H_{40}$? Wait, no—wait, the question says "the substance composed of 10 such molecules" (assuming "such" refers to ethylene). Wait, maybe a typo? Wait, original problem: "the chemical formula for the substance composed of 10 such molecules" (ethylene, $C_2H_4$). So 10 molecules of $C_2H_4$ would be $10C_2H_4$, but if it’s a single formula (like a polymer? No, 10 molecules of ethylene). Wait, maybe the options? Wait, the given options: $C_2H_4$, $CH_4$, $C_{10}H_20$? Wait, no—wait, the user’s image shows "C₂H₄", "CH₄", "C₁₀H₂₀"? Wait, maybe the question is about the formula for ethylene (1 molecule) or 10 molecules. Wait, ethylene’s formula is $C_2H_4$. If 10 molecules, it’s $10C_2H_4$, but maybe the question is misphrased. Alternatively, maybe it’s a polymer, but no—10 molecules of ethylene would be $10C_2H_4$. But if the options include $C_{10}H_{20}$, that’s wrong. Wait, maybe the question is "the chemical formula for the substance (ethylene) composed of 10 such molecules"—no, ethylene is $C_2H_4$ per molecule. Wait, maybe the answer is $C_2H_4$ (for one molecule) or $10C_2H_4$. But given the options, $C_2H_4$ is ethylene’s formula.
Step-by-Step:
Step 1: Identify Ethylene’s Formula
Ethylene (ethene) has 2 C and 4 H per molecule: $C_2H_4$.
Step 2: Formula for 10 Molecules
For 10 molecules, the formula is $10 \times C_2H_4 = 10C_2H_4$. But if the question means the empirical formula or molecular formula of the substance (assuming 10 molecules as a unit), but typically, molecular formula for ethylene is $C_2H_4$. If the options include $C_2H_4$, that’s the answer.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Left model (labeled "ethylene" or "ethene")