QUESTION IMAGE
Question
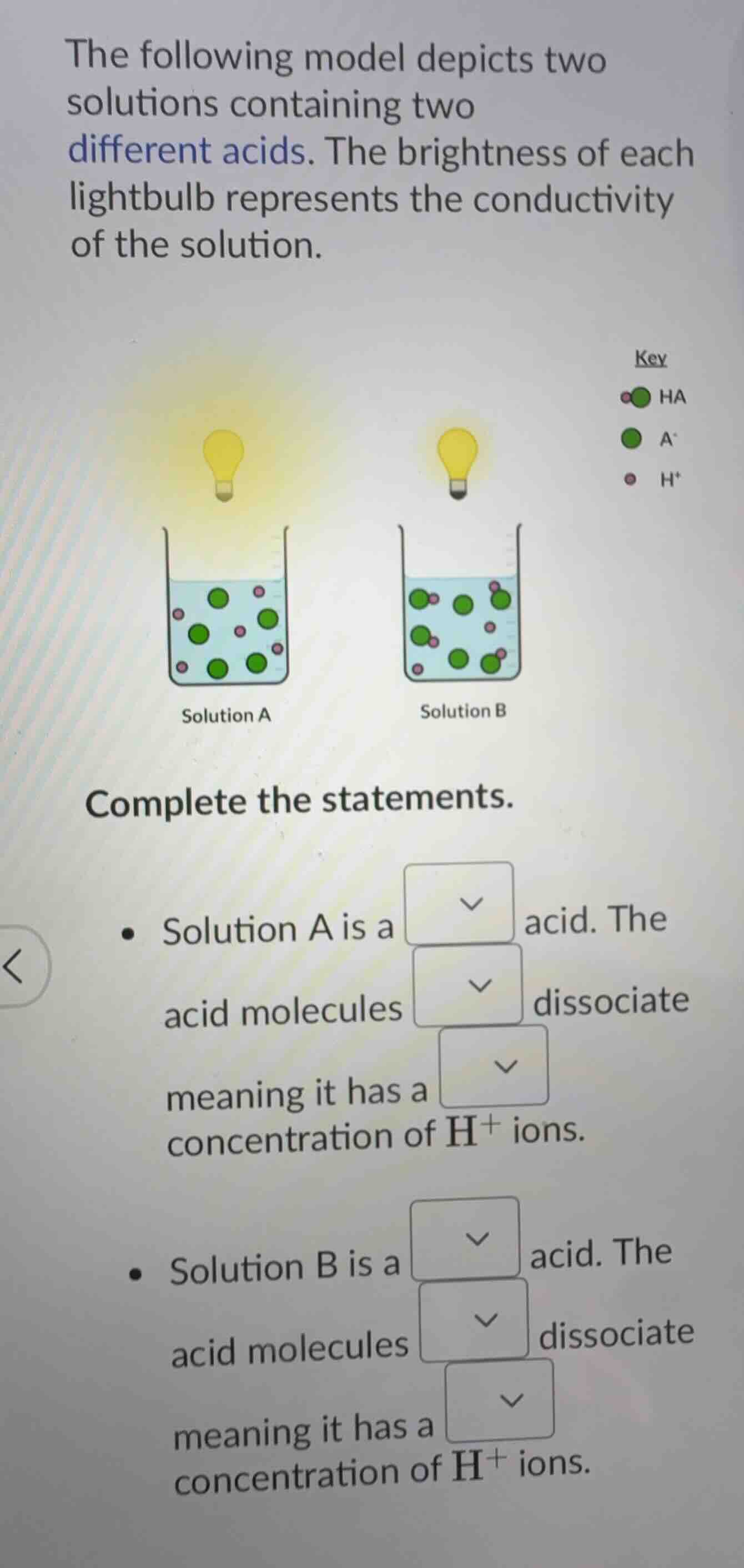
the following model depicts two solutions containing two different acids. the brightness of each lightbulb represents the conductivity of the solution. complete the statements. - solution a is a dropdown acid. the acid molecules dropdown dissociate meaning it has a dropdown concentration of h⁺ ions. - solution b is a dropdown acid. the acid molecules dropdown dissociate meaning it has a dropdown concentration of h⁺ ions.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Solution A is a strong acid. The acid molecules completely dissociate meaning it has a high concentration of $\text{H}^+$ ions.
Solution B is a weak acid. The acid molecules partially dissociate meaning it has a low concentration of $\text{H}^+$ ions.